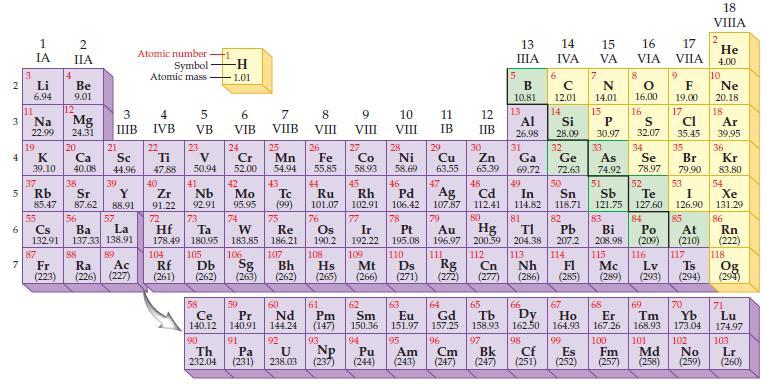
Refer to the periodic table and predict the number of valence electrons for an atom of each
Question:
Refer to the periodic table and predict the number of valence electrons for an atom of each of the following representative elements:
(a) Na
(b) Al
(c) S
(d) Xe.
Periodic Table:
Transcribed Image Text:
2 3 4 5 N.. 3 7 Li 6.94 11 1 IA Na 22.99 19 37 G 85.47 4 2 IIA Be 9.01 38 Rb Sr 87 12 K Ca Sc 39.10 40.08 44.96 87.62 56 55 6 Cs Ba Fr (223) Mg 24.31 20 21 88 39 3 IIIB Y 88.91 57 Lo 132.91 137.33 138.91 La 89 O Atomic number Symbol Atomic mass Ra Ac (226) (227) 4 IVB 22 Ti 47.88 40 Zr 91.22 72 5 VB 23 V 50.94 41 Nb 92.91 73 Hf Ta 178.49 180.95 104 105 ᎠᏏ Rf (261) (262) 58 Ce 140.12 90 Th 232.04 -H 1.01 6 VIB 24 Cr 52.00 42 Mo 95.95 74 W 183.85 7 VIIB 91 25 Pa (231) Mn 54.94 43 Tc (99) 75 59 60 Pr Nd 140,91 144.24 Re 186.21 8 VIII 92 U 238.03 26 Fe 55.85 44 Ru 101.07 76 Os 190.2 108 9 VIII 27 Np (237) Co 58.93 77 78 Ir 192.22 109 Mt Pt 195.08 110 Ds 106 107 Hs Rg Sg Bh Cn (263) (262) (265) (266) (271) (272) (277) 10 11 VIII IB 28 Ni 58.69 45 46 Rh Pd 102.91 106.42 61 62 63 Pm Sm Eu (147) 150.36 151.97 93 94 95 Pu Am (244) (243) 29 Cu 63.55 47 12 IIB 30 111 Zn 65.39 5 13 ΠΙΑ B 10.81 13. Al 26.98 31 Ga 69.72 48 Ag 107.87 79 Cd 112.41 80 82 Au Hg Pb 196.97 200.59 204.38 207.2 112 113 114 Nh FI (286) (285) 49 In 114.82 81 14 IVA F 7 6 C 12.01 14 15 Se 50 Si P 28.09 30.97 32 33 Ge As 72.63 74.92 78.97 51 52 Sn Sb Te 118.71 121.75 127.60 83 84 Bi Po 208.98 (209) 115 116 Mc Lv Ts (289) (293) (294) 64 66 67 65 Gd Tb Dy Ho 157.25 158.93 162.50 164.93 96 97 98 99 Cm Bk Cf (247) (247) (251) 15 VA N 14.01 16 17 VIA VIIA 68 Er 167.26 100 Es Fm (252) (257) 8 9 0 F 16.00 19.00 17 16 S 32.07 34 cl 35.45 35 Br 79.90 53 I 126.90 85 At (210) 117 69 70 Tm Yb 168.93 173.04 101 102 Md No (258) (259) 18 VIIIA 2 He 4.00 10 Ne 20.18 18 Ar 39.95 36 Kr 83.80 54 Xe 131.29 86 Rn (222) 118 Og (294) 71 Lu 174.97 103 Lr (260)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (1 review)
Find the element in the periodic table note the group number and indicate the number of valence elec...View the full answer

Answered By

SUMAN DINDA
I LIKE TO TEACH STUDENTS. SO, I START MYSELF AS A PRIVATE TUTOR. I TEACH STUDENTS OF DIFFERENT CLASSES. I HAVE ALSO DONE BACHELOR OF EDUCATION DEGREE(B.ED). DURING THIS COURSE I HAD TO TEACH IN A SCHOOL. SO I HAVE A GOOD EXPERIENCE IN TEACHING.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Introductory Chemistry Concepts And Critical Thinking
ISBN: 9780321804907
7th Edition
Authors: Charles Corwin
Question Posted:
Students also viewed these Sciences questions
-
The electronics industry manufactures transistors using arsenic diffusion. Refer to the periodic table and predict an element that may substitute for arsenic. 2 3 4 5 6 7 3 11 1 IA Li Na 19 37 Rb 55...
-
The electronics industry manufactures semiconductor chips from silicon. Refer to the periodic table and predict an element that may substitute for silicon. 2 3 4 5 6 7 3 11 1 IA Li Na 19 37 Rb 55 87...
-
Refer to the periodic table and predict the number of 5d electrons in a Pt atom. Periodic Table: 2 3 4 10 6 3 7 11 Li 6.94 1 IA Na 22.99 19 37 5 Rb K 39.10 al 55 87 4 Fr (223) 2 IIA Be 9.01 12 Mg...
-
1:When developing a marketing strategy for business customers, it is essential to understand the process the business goes through when making a buying decision. Knowledge of business buying behavior...
-
Breyer Memorial Hospital received a $100,000 gift that was restricted by the donor for heart research. At fiscal year-end Breyer had incurred $25,000 in expenses related to this project. Explain how...
-
On January 1, 2011, Randy Incorporated purchased $500,000 of 20-year, 10% bonds when the market rate of interest was 8%. Interest is to be paid on June 30 and December 31 of each year. 1. Prepare the...
-
Select a publicly traded company for analysis or use a company assigned by your instructor. Based on the firms most recent Form 10-K report (accessed through the SEC EDGAR database or from the...
-
As you learned earlier in this chapter, R/Way is a small but rapidly growing trucking company headquartered in Cleveland, Ohio. R/Ways information system currently consists of a file server and three...
-
There is an 11% chance that a booked gig is cancelled. If you have 4 booked gigs in a row, what is the probability that none of them get cancelled? Round the answer to the nearest 0.1%.
-
What is the electron dot formula for an atom of neon? (a) (b) (c) (d) (e) Ne
-
Predict the number of valence electrons for a Group VIIA/17 element. (a) 1 (b) 2 (c) 3 (d) 7 (e) 17.
-
Think about a recent presentation you attended that you found effective. In three to five paragraphs, describe why it was effective. Include the following aspects in your analysis, referring to...
-
Construct a 95 % confidence interval for the mean home price. What happens to the size of the confidence interval?
-
Suppose X has an exponential distribution with = 1/3 . Find the following probabilities: (a) P(3 < X < 5) (b) P(5 X 10) (c) P(2 < X < 1)
-
Go to the St. Louis Federal Reserve FRED database and find data on the net saving rate as a percentage of national income (W207RC1A156NBEA). a) Calculate the average net saving rate over the period...
-
Suppose X has an exponential distribution with = 4. Find the following probabilities: VI VI VI (a) P(X .3) (b) P(X < .5) (c) P(X 1.6)
-
A bank manager finds from reviewing her records that the amount of money deposited on Saturday morning is normally distributed with a standard deviation of $150. A random sample of 7 customers...
-
Develop a multiple regression model to predict y from x1, x2, and x3 using the following data. Discuss the values of F andt. 201 194238676138215 01965685004242 423344332322344 C.| 1 0 3 8 3 5 4 4 2 9...
-
SBS Company have received a contract to supply its product to a Health Care Service Hospital. The sales involve supplying 1,250 units every quarter, the sales price is RM 85 per unit. The Client...
-
The board of directors of Gifford Corp. declared cash dividends of $260,000 during the current year. If dividends payable was $85,000 at the beginning of the year and $90,000 at the end of the year,...
-
Explain how the amount of cash payments to suppliers is computed under the direct method.
-
The net income for Letterman Company for 2010 was $320,000. During 2010, depreciation on plant assets was $124,000, amortization of patent was $40,000, and the company incurred a loss on sale of...
-
What is the Present Value for $15,000 in 30 years at discount rate of 12%? XYZ mutual fund has an average 6% annual rate of return. How much money you could make in 20 years if you invest 80,000 at...
-
1. What is a credit card, and how does it work? 2. What factors should individuals consider when choosing a credit card? 3. How does interest accrue on credit card balances, and what is the typical...
-
Assume that the First StatBaxter Equipment earned $300,000 last year. Its owners' equity totaled $2,500,000. Based on these amounts, what is the firm's return on owners' equity?

Study smarter with the SolutionInn App


