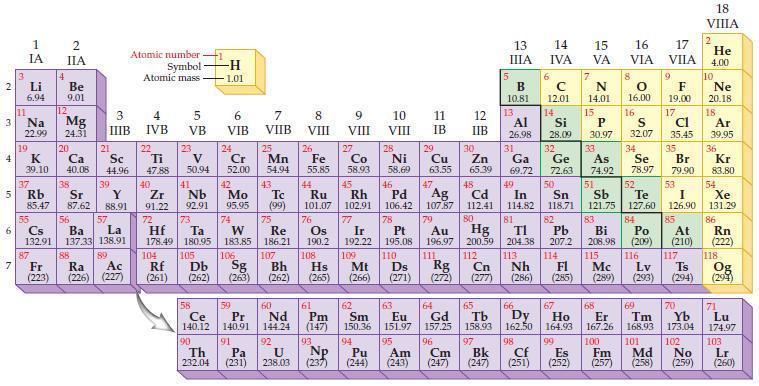
Refer to the periodic table and select the symbol of the element that fits each of the
Question:
Refer to the periodic table and select the symbol of the element that fits each of the following descriptions.
(a) The semimetal in the second period
(b) The semimetal in the fourth period and Group IVA/14
(c) The halogen that exists as yellow gas
(d) The halogen that is radioactive.
Periodic Table:
Transcribed Image Text:
2 3 4 10 6 3 7 11 Li 6.94 1 IA Na 22.99 19 37 5 Rb K 39.10 al 55 4 87 2 IIA Fr (223) Be 9.01 12 Mg 24.31 Sr 85.47 87.62 20 38 Ca Sc 40.08 44.96 21 56 Cs La Ba 132.91 137.33 138.91 88 3 IIIB 39 Y 88.91 57 89 Atomic number Symbol Atomic mass Ra Ac (226) (227) 4 IVB 22 Ti 47.88 40 Zr 91.22 72 5 VB 23 V 50.94 41 Nb 92.91 73 -H 1.01 6 VIB 58 Ce 140.12 90 Th 232.04 24 Cr 52.00 42 Mo 95.95 74 104 105 Rf Db (261) (262) (263) Sg 106 59 Pr 140.91 91 7 VIIB Hf Ta W Re 178.49 180.95 183.85 186.21 Pa (231) 25 Mn 54.94 43 Tc (99) 75 107 60 Nd 144.24 8 VIII 92 26 Fe Co 55.85 58.93 Os 190.2 44 45 Ru Rh 101.07 102.91 76 9 VIII 61 Pm (147) 27 108 Bh Mt Hs (262) (265) (266) 93 U NP 238.03 (237) 77 Ir 192.22 109 10 VIII 62 Sm 150.36 94 28 Ni 58.69 46 Pd 106.42 78 11 IB 12 IIB 13 IIIA 5 B 10.81 13 Al 26.98 14 15 IVA 6 C 12.01 14 Si 28.09 32 17 16 VA VIA VIIA 8 7 N 14.01 15 P 30.97 79 Pt 195.08 110 Ds 81 82 Au Hg TI Pb 196.97 200.59 204.38 207.2 111 112 113 114 Rg Cn Nh Fl (271) (272) (277) (286) (285) 29 30 31 33 As Ge Se Cu Zn Ga 63.55 65.39 69.72 72.63 74.92 78.97 16.00 16 S 32.07 34 47 51 48 49 50 52 Ag Cd In Sn Sb Te 107.87 112.41 114.82 118.71 121.75 127.60 80 83 Bi 208.98 115 84 Po (209) 9 F 19.00 Md (258) 17 Cl 35.45 35 Br 79.90 53 I 126.90 85 At (210) 117 116 Mc Ts Lv (289) (293) (294) 66 67 70 63 64 65 68 69 Eu Gd Tb Dy Но Er Tm Yb 151.97 157.25 158.93 162.50 164.93 167.26 168.93 173.04 96 98 99 100 Cf Es Fm (251) (252) (257) 95 101 102 Pu Am (244) (243) Cm (247) 97 Bk (247) 18. VIIIA 2 He 4.00 10 Ne 20.18 18 Ar 39.95 36 Kr 83.80 54 Xe 131.29 86 Rn (222) 118 Og (294) 71 Lu 174.97 103 No Lr (259) (260)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (3 reviews)
Element Symbols based on Descriptions a Boron B This is the only semimetal in the second pe...View the full answer

Answered By

Shehar bano
I have collective experience of more than 7 years in education. my area of specialization includes economics, business, marketing and accounting. During my study period I remained engaged with a business school as a visiting faculty member and did a lot of business research. I am also tutoring and mentoring number of international students and professionals online for the last 7 years.
5.00+
4+ Reviews
10+ Question Solved
Related Book For 

Introductory Chemistry Concepts And Critical Thinking
ISBN: 9780321804907
7th Edition
Authors: Charles Corwin
Question Posted:
Students also viewed these Sciences questions
-
Refer to the periodic table and select the symbol of the element that fits each of the following descriptions. (a) The semimetal in the third period (b) The semimetal in the fourth period and Group...
-
Refer to the periodic table and select the symbol of the element that fits each of the following descriptions. (a) The fourthperiod alkali metal (b) The fourthperiod alkaline earth metal (c) The rare...
-
Refer to the periodic table and select the symbol of the element that fits each of the following descriptions. (a) The thirdperiod alkali metal (b) The thirdperiod alkaline earth metal (c) The rare...
-
Determine if the following strains satisfy the compatibility equations (2.6.2): a. b. c. where A, B and C are constants. Equation 2.6.2 ex=Ay, ey = ez = 0, exy = (Ax+Bz)/2, eyz = Bxz + Cy, ezx = C.x
-
Single Audit. Background, Mountain Lake Mental Health Affiliates, a nongovernmental not-for-profit organization, has contacted Rill Wise, CPA, about conducting an annual audit for its first year of...
-
The income statement for Fignon Co. for the year ended December 31, 2011, reported the following. Income from continuing operations before income taxes . . . . . . . . . . . . . . . . . . . . . . . ....
-
JDLs inventory turnover is: (a) 1.25 times. (b) 13.5 times. (c) 3.0 times. (d) 37.5 times. JDL Corporation Selected Financial Data, December 31, 2024 Current assets $150,000 Current liabilities...
-
The Mason Corporations present capital structure, which is also its target capital structure, calls for 50 percent debt and 50 percent common equity. The firm has only one potential project, an...
-
Solve the equation. Check the solution. 4 (2x-1)=-1-1 7 7
-
Identify the group number corresponding to each of the following families of elements. (a) Boron group (b) Oxygen group (c) Nickel group (d) Copper group.
-
Identify the group number corresponding to each of the following families of elements. (a) Alkali metals (b) Alkaline earth metals (c) Halogens (d) Noble gases.
-
A discrete random variable x can assume five possible values: 2, 3, 5, 8, and 10. Its probability distribution is shown here: a. What is p(5)? b. What is the probability that x equals 2 or 10? c....
-
What is the difference between the short run and the long run in macroeconomic analysis? Why do macroeconomists differentiate between the two time horizons?
-
In the per-worker production function, what factors determine the level of output per worker? Which one of these factors does the Solow growth model consider to be exogenous?
-
How do you participate in the debate on state ownership versus private ownership?
-
As a manager, is it ethical to threaten your suppliers? Your buyers?
-
Beginning from a steady state in the Solow growth model, explain how an increase in the saving rate will affect the levels and growth rates of capital and output per worker.
-
The U.S. Commodities Futures Trading Commission reports on the volume of trading in the U.S. commodity futures exchanges. Shown here are the figures for grain, oilseeds, and livestock products over a...
-
Nitrogen monoxide reacts with hydrogen as follows: 2NO(g)+ H2(g) N2O(g) + H2O(g) The rate law is [H2]/ t = k[NO]2[H2], where k is 1.10 107 L2/(mol2s) at 826oC. A vessel contains NO and H2 at...
-
(Preparation of Pension Worksheet) Using the information in E20-2 prepare a pension worksheet inserting January 1, 2010, balances, showing December 31, 2010, balances, and the journal entry recording...
-
(Basic Pension Worksheet) The following facts apply to the pension plan of Boudreau Inc. for the year 2010. Using the preceding data, compute pension expense for the year 2010, as part of your...
-
(Application of Years-of-Service Method) Andrews Company has five employees participating in its defined-benefit pension plan. Expected years of future service for these employees at the beginning of...
-
On March 13, 2020, you convert 500,000 U.S. dollars to Japanese yen in the spot foreign exchange market and purchase a six-month forward contract to convert yen into dollars. How much will you...
-
When applying for Medicaid all of the following constitute a "countable asset" EXCEPT: Spouse's section 401(k) plan Equity in home owned jointly with spouse Worker's own section 401(k) plan Jointly...
-
You have just graduated from university and are considering starting a new line clothing. To start the new line of clothing you require a capital outlay of $25,000 and net working capital of $1,000....

Study smarter with the SolutionInn App


