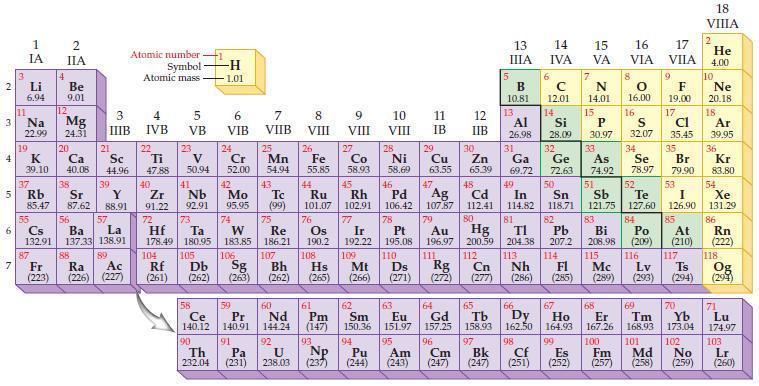
Refer to the periodic table and select the symbol of the element that fits each of the
Question:
Refer to the periodic table and select the symbol of the element that fits each of the following descriptions.
(a) The fourth–period alkali metal
(b) The fourth–period alkaline earth metal
(c) The rare earth element with the lowest atomic mass
(d) The lanthanide that does not occur naturally.
Periodic Table:
Transcribed Image Text:
2 3 4 10 6 3 7 11 Li 6.94 1 IA Na 22.99 19 37 5 Rb K 39.10 al 55 4 87 2 IIA Fr (223) Be 9.01 12 Mg 24.31 Sr 85.47 87.62 20 38 Ca Sc 40.08 44.96 21 56 Cs La Ba 132.91 137.33 138.91 88 3 IIIB 39 Y 88.91 57 89 Atomic number Symbol Atomic mass Ra Ac (226) (227) 4 IVB 22 Ti 47.88 40 Zr 91.22 72 5 VB 23 V 50.94 41 Nb 92.91 73 -H 1.01 6 VIB 58 Ce 140.12 90 Th 232.04 24 Cr 52.00 42 Mo 95.95 74 104 105 Rf Db (261) (262) (263) Sg 106 59 Pr 140.91 91 7 VIIB Hf Ta W Re 178.49 180.95 183.85 186.21 Pa (231) 25 Mn 54.94 43 Tc (99) 75 107 60 Nd 144.24 8 VIII 92 26 Fe Co 55.85 58.93 Os 190.2 44 45 Ru Rh 101.07 102.91 76 9 VIII 61 Pm (147) 27 108 Bh Mt Hs (262) (265) (266) 93 U NP 238.03 (237) 77 Ir 192.22 109 10 VIII 62 Sm 150.36 94 28 Ni 58.69 46 Pd 106.42 78 11 IB 12 IIB 13 IIIA 5 B 10.81 13 Al 26.98 14 15 IVA 6 C 12.01 14 Si 28.09 32 17 16 VA VIA VIIA 8 7 N 14.01 15 P 30.97 79 Pt 195.08 110 Ds 81 82 Au Hg TI Pb 196.97 200.59 204.38 207.2 111 112 113 114 Rg Cn Nh Fl (271) (272) (277) (286) (285) 29 30 31 33 As Ge Se Cu Zn Ga 63.55 65.39 69.72 72.63 74.92 78.97 16.00 16 S 32.07 34 47 51 48 49 50 52 Ag Cd In Sn Sb Te 107.87 112.41 114.82 118.71 121.75 127.60 80 83 Bi 208.98 115 84 Po (209) 9 F 19.00 Md (258) 17 Cl 35.45 35 Br 79.90 53 I 126.90 85 At (210) 117 116 Mc Ts Lv (289) (293) (294) 66 67 70 63 64 65 68 69 Eu Gd Tb Dy Но Er Tm Yb 151.97 157.25 158.93 162.50 164.93 167.26 168.93 173.04 96 98 99 100 Cf Es Fm (251) (252) (257) 95 101 102 Pu Am (244) (243) Cm (247) 97 Bk (247) 18. VIIIA 2 He 4.00 10 Ne 20.18 18 Ar 39.95 36 Kr 83.80 54 Xe 131.29 86 Rn (222) 118 Og (294) 71 Lu 174.97 103 No Lr (259) (260)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 83% (6 reviews)
a The fourthperiod alkali metal K Potassium ...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Introductory Chemistry Concepts And Critical Thinking
ISBN: 9780321804907
7th Edition
Authors: Charles Corwin
Question Posted:
Students also viewed these Sciences questions
-
Refer to the periodic table and select the symbol of the element that fits each of the following descriptions. (a) The thirdperiod alkali metal (b) The thirdperiod alkaline earth metal (c) The rare...
-
Refer to the periodic table and select the symbol of the element that fits each of the following descriptions. (a) The semimetal in the third period (b) The semimetal in the fourth period and Group...
-
Refer to the periodic table and select the symbol of the element that fits each of the following descriptions. (a) The semimetal in the second period (b) The semimetal in the fourth period and Group...
-
A Canadian packaging company wished to extend its activities in the area of convenience foods. It had pinpointed one particular area where it could supply pizza boxes to half a dozen chains of pizza...
-
Multiple Choice. Choose the best answer. 1. Which of the following activities would always indicate that an auditors independence has been impaired? a. Providing advice on establishing an internal...
-
A small fraction of the energy in a sound wave is absorbed by the air through which the sound passes. How does this modify the inverse square relationship between intensity and distance from the...
-
What is the P/D ratio? How may it be improved? LO.1
-
Consider the intersection of two shocks of opposite families, as sketched in Fig. 4.23. For M 1 = 3, p 1 = 1 atm, ? 2 = 20?, and ? 3 = 15?, calculate the pressure in regions 4 and 4?, and the flow...
-
A restaurant has three main products. Fixed costs are $397,000 and the tax rate is 25%. Other financial information is as follows: Item Average Purchase (in $) Average Margin Total Sales (in %)...
-
According to the general trend, metallic character (increases/decreases) proceeding up a group of elements in the periodic table. Periodic Table: 2 3 4 10 6 3 7 11 Li 6.94 1 IA Na 22.99 19 37 5 Rb K...
-
According to the American convention, what is the designation for each of the following groups of elements? (a) Group 2 (b) Group 4 (c) Group 6 (d) Group 8 (e) Group 11 (f) Group 12 (g) Group 16 (h)...
-
Repeat Example 14-2 except for a single-stage system and unknown underflow product concentration. Example 14-2 We wish to treat 1000.0 kg/h (wet basis) of insoluble meal (D) that contains 20.0 wt%...
-
In this scenario you are the Manager of a Home and Community Care organization in Victoria. Your organization provides oversees support services for individuals in their homes. The individuals that...
-
find the wind speed and direction for the following parcels. Important note: To determine the arc tangent using the Google calculator, click the box marked "Inv" so that it is a light gray instead of...
-
As consumers we're connected to an instant feed or live updates, breaking news, and messages. We believe that when we post something on social media, we will get instant feedback from friends. What...
-
Describe the process for screening candidates for ethics. Outline which job candidate factors are illegal to consider when hiring. Explain how to obtain accurate behavior information from resumes,...
-
In relation to Operation management, sustainability and supply chain management explain the following terms; What is the overall objective of scheduling? What is JIT? What is a Lean producer? Discuss...
-
What is the amount of the permitted deduction for single and married taxpayers who use Form 1040EZ?
-
Write a program to move a signed number from smaller register to bigger register. Hint: movzx ax, bl Topic: Data Related Operators and Directives in assembly language
-
Access the glossary (Master Glossary) to answer the following. (a) What is an accumulated benefit obligation? (b) What is a defined benefit postretirement plan? (c) What is the definition of...
-
In general, how can an employer choose an appropriate discount rate for its pension plan? What information could an employer use in choosing a discount rate?
-
If an employer has a defined-benefit pension plan, what components would make up its net periodic pension cost?
-
1,600 Balance Sheet The following is a list (in random order) of KIP International Products Company's December 31, 2019, balance sheet accounts: Additional Paid-In Capital on Preferred Stock $2,000...
-
Question 3 4 pts 9 x + 3 x 9 if x 0 Find a) lim f(x), b) lim, f(x), C), lim , f(x) if they exist. 3 Edit View Insert Format Tools Table : 12pt M Paragraph B IV A2 Tv
-
Mr. Geoffrey Guo had a variety of transactions during the 2019 year. Determine the total taxable capital gains included in Mr. Guo's division B income. The transactions included: 1. On January 1,...

Study smarter with the SolutionInn App


