Refer to the periodic table and state the mass of 6.02 x 10 23 atoms of each
Question:
Refer to the periodic table and state the mass of 6.02 x 1023 atoms of each of the following nonmetals.
(a) Beryllium
(b) Barium
(c) Boron
(d) Bromine.
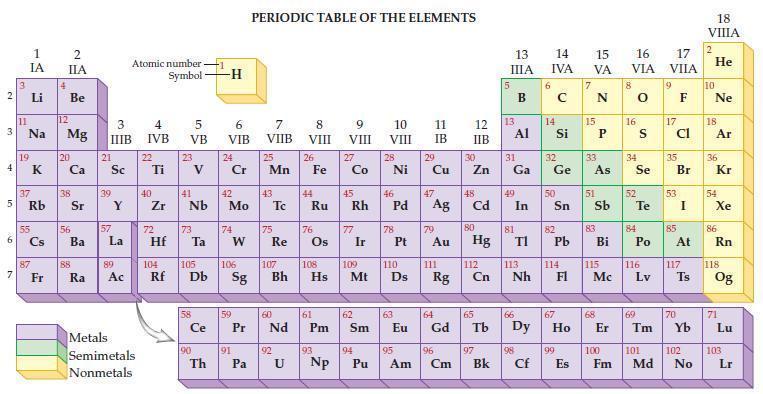
Transcribed Image Text:
2 3 4 AD 6 3 7 11 1 IA 37 5 Rb 4 Li B Na 19 55 87 2 IIA Fr Be 12 Mg 20 38 56 Ba 88 Ra 3 IIIB 21 Sc 39 57 Y La 89 Atomic number Symbol Ac Metals Semimetals Nonmetals 4 IVB 22 Ti 40 Zr 5 VB 72 23 41 Nb 73 Hf Ta 104 105 Rf Db 90 -H Th 6 VIB 24 Cr 42 Mo 74 W 106 91 PERIODIC TABLE OF THE ELEMENTS 7 VIIB Pa 25 Mn 43 59 60 Ce Pr Nd 75 Re 92 8 VIII U 26 107 108 Sg Bh Hs 44 Fe Ru 76 61 Pm 93 Np 9 VIII 27 Co 45 Rh 77 Ir 109 62 Sm 94 10 VIII 110 Mt Ds Pu 28 Ni 78 63 46 47 48 Pd Ag Cd 11 IB 95 Am 29 30 Cu Zn 79 Au 64 Eu Gd 111 Rg 12 IIB 96 Cm 80 Hg 112 Cn 65 97 Bk 13 IIIA 13 Al 31 2 Ga 49 81 In TI 113 66 Nh 98 Cf 14 15 16 IVA VA VIA 6 14 32 Ge 50 82 Pb 114 33 e As 67 99 E Es 51 83 Bi 115 Mc 68 8 16 34 Se 52 S Te 84 Po 116 Lv 69 17 VIIA 9 17 35 Br 53 85 At 117 Ts 70 18 VIIIA 2 He 10 Ne 18 Ar 36 54 Kr Xe 86 Rn 118 Og 71 Lu 100 101 102 103 Fm Md No Lr
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
To find the mass of 602 x 1023 atoms of each element well need their respective ...View the full answer

Answered By

Ali Khawaja
my expertise are as follows: financial accounting : - journal entries - financial statements including balance sheet, profit & loss account, cash flow statement & statement of changes in equity -consolidated statement of financial position. -ratio analysis -depreciation methods -accounting concepts -understanding and application of all international financial reporting standards (ifrs) -international accounting standards (ias) -etc business analysis : -business strategy -strategic choices -business processes -e-business -e-marketing -project management -finance -hrm financial management : -project appraisal -capital budgeting -net present value (npv) -internal rate of return (irr) -net present value(npv) -payback period -strategic position -strategic choices -information technology -project management -finance -human resource management auditing: -internal audit -external audit -substantive procedures -analytic procedures -designing and assessment of internal controls -developing the flow charts & data flow diagrams -audit reports -engagement letter -materiality economics: -micro -macro -game theory -econometric -mathematical application in economics -empirical macroeconomics -international trade -international political economy -monetary theory and policy -public economics ,business law, and all regarding commerce
4.00+
1+ Reviews
10+ Question Solved
Related Book For 

Introductory Chemistry Concepts And Critical Thinking
ISBN: 9780321804907
7th Edition
Authors: Charles Corwin
Question Posted:
Students also viewed these Sciences questions
-
Refer to the periodic table and state the mass of 6.02 x 10 23 atoms of each of the following metals. (a) Sodium (b) Strontium (c) Silicon (d) Selenium. 2 3 4 av 6 3 7 11 1 IA 37 5 Rb 4 Li B Na 19 55...
-
Refer to the periodic table and state the mass for each of the following number of atoms. (a) 1 atom of carbon (b) 6.02 x 10 23 atoms of carbon. 2 3 4 av 6 3 7 11 1 IA 37 5 Rb Li Na 19 55 87 Fr 4 2...
-
What would you suggest to be done and which principles are important to you in this decision? How would you think about the greatest good in this case? As a leader what are your duties and who are...
-
Describe the three ways a client can reference a name from a namespace in C++.
-
The balance sheet for Focus Consulting reports the following information on July 1,2012. Focus decides to redeem these bonds at 101 after paying semiannual interest. Prepare the journal entry to...
-
What sets Oakland Athletics coach Billy Beane apart from other coaches and scouts in baseball?
-
The current loop in Figure P28.34 lies in the \(x y\) plane. For each of the Amperrian paths (a)-(e), is the line integral of the magnetic field positive, negative, or zero? Data from Figure P28.34...
-
During its first year of operations, Rosa Corporation had these transactions pertaining to its common stock . Jan. 10 Issued 30,000 shares for cash at $5 per share. July 1 Issued 60,000 shares for...
-
Malimali Fish Products Ltd, A Malindi-based firm dealing in fish products, conducts training programmes for all its line managers. The managers ar expected to master, in particular, procedures in...
-
What is the number of molecules in 1.00 mol of a diatomic nonmetal?
-
Calculate the number of moles of potassium in 1.25 x 10 21 atoms K. Strategy Plan STEP 1: What unit is asked for in the answer? STEP 2: What given value is related to the answer? STEP 3: What unit...
-
The Black Knights Inc., a manufacturer of low-sugar, low-sodium, low-cholesterol TV dinners, would like to increase its market share in Western Canada. In order to do so, Black Knights has decided to...
-
If a CD manufacturing company produces a variety of different CDs for computers. The CDs are produced at a rate of 200,000 per day and shipped out at a rate of 20,000 per day. The CDs are produced in...
-
Use the currency converter. In your answers, record only the numbers in bold text. Do not round up or down. For example, if the answer is 46.79 552, then record only 46.79. 1 U.S. Dollar =...
-
How would one describe Huntley school distirct 158 current financial status?
-
Since he was 25 years old, Ben has been depositing $250 at the end of each month into a tax-free retirement account earning interest at the rate of 5.5%/year compounded monthly. Larry, who is the...
-
1. According to Erikson, young adults must resolve the psychosocial conflict of. 2. Why are formal rites of passage important? What has Western society lost by eliminating them? What has it gained?...
-
Jared Monsma, Weekend Golfers vice president for marketing, has concluded from his market analysis that sales have been dwindling for the standard golf cart because of aggressive pricing by...
-
What does non-recourse financing mean?
-
Navajo Corporation traded a used truck (cost $20,000, accumulated depreciation $18,000) for a small computer worth $3,300. Navajo also paid $500 in the transaction. Prepare the journal entry to...
-
Use the information for Navajo Corporation from BE10-8. Prepare the journal entry to record the exchange, assuming the exchange lacks commercial substance.
-
Mehta Company traded a used welding machine (cost $9,000, accumulated depreciation $3,000) for office equipment with an estimated fair value of $5,000. Mehta also paid $3,000 cash in the transaction....
-
A figure skater spins at the end of her routine and slows down with an angular acceleration of "0.4" A per second squared. If she initially spun with a frequency of 2.35 Hz, how much time does it...
-
2 Charlie's Crispy Chicken (CCC) operates a fast-food restaurant. When accounting for its first year of business, CCC created several accounts. Account Name Description Balance Accounts Payable...
-
Would you make any changes to the Federal tax policy related to this area of the IRC ? If so , what would you change and why? How would these changes help rectify the disparate outcomes by race?

Study smarter with the SolutionInn App


