Refer to the periodic table and state the mass for each of the following number of atoms.
Question:
Refer to the periodic table and state the mass for each of the following number of atoms.
(a) 1 atom of carbon
(b) 6.02 x 1023 atoms of carbon.
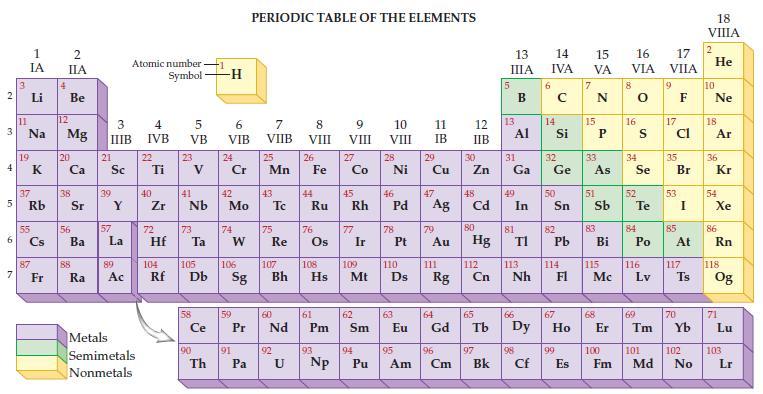
Transcribed Image Text:
2 3 4 av 6 3 7 11 1 IA 37 5 Rb Li Na 19 55 87 Fr 4 2 IIA Be 12 Mg 20 38 56 Ba 88 Ra 3 IIIB 21 Sc 39 57 Y La 89 Ac Atomic number Symbol Metals Semimetals Nonmetals 4 IVB 22 Ti 40 Zr 5 VB 72 23 41 Nb 73 Hf Ta Ce 90 -H Th 6 VIB 24 Cr 42 Mo 74 W 59 91 PERIODIC TABLE OF THE ELEMENTS 7 VIIB Pa 25 Mn 43 75 60 Pr Nd Re 92 8 VIII U 26 44 Fe Ru 76 61 104 105 106 107 108 109 110 Rf Db Sg Bh Hs Mt D Pm 93 9 VIII Np 27 Co 77 Ir 62 Sm 10 VIII 45 46 47 Rh Pd Ag 94 28 Pu Ni 78 63 11 IB 29 95 Cu 79 Au 111 64 Eu Gd Rg 96 Am Cm 12 13 IIB 30 Zn 48 Cd 80 Hg 112 Cn 65 97 13 IIIA Bk 31 Al Ga 49 In 81 TI 113 Nh 66 98 Cf 14 15 16 IVA VA VIA 6 14 50 82 Pb 32 33 34 Ge As Se 114 67 99 E 15 Es 51 83 Bi 115 Mc 68 8 16 52 S Te 84 Po 116 Lv 69 17 VIIA 9 17 cl 35 Br 53 85 At 117 Ts 70 100 101 102 Fm Md No 18 VIIIA 2 He 10 Ne 18 Ar 36 Kr 54 Xe 86 Rn 118 Og 71 Lu 103 Lr
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
a A carbon atom has ...View the full answer

Answered By

Susan Juma
I'm available and reachable 24/7. I have high experience in helping students with their assignments, proposals, and dissertations. Most importantly, I'm a professional accountant and I can handle all kinds of accounting and finance problems.
4.40+
15+ Reviews
45+ Question Solved
Related Book For 

Introductory Chemistry Concepts And Critical Thinking
ISBN: 9780321804907
7th Edition
Authors: Charles Corwin
Question Posted:
Students also viewed these Sciences questions
-
Refer to the periodic table and state the mass of 6.02 x 10 23 atoms of each of the following metals. (a) Sodium (b) Strontium (c) Silicon (d) Selenium. 2 3 4 av 6 3 7 11 1 IA 37 5 Rb 4 Li B Na 19 55...
-
Refer to the periodic table and state the mass of 6.02 x 10 23 atoms of each of the following nonmetals. (a) Beryllium (b) Barium (c) Boron (d) Bromine. 2 3 4 AD 6 3 7 11 1 IA 37 5 Rb 4 Li B Na 19 55...
-
Refer to the periodic table and state the number of atoms for each of the following nonmetals. (a) 30.97 g P (b) 20.18 g Ne. Periodic Table: 19 2 3 4 5 6 7 3 11 1 IA Li 6.94 Na 22.99 19 37 4 12 87 2...
-
One end of a light, elastic string, of natural length 1.2m and modulus of elasticity 32N, is attached to a fixed point, B. A particle, P, of mass 1.5 kg, is then attached to the other end of the...
-
Alvarez Company has the following data for the weekly payroll ending January 31. Employees are paid 1 times the regular hourly rate for all hours worked in excess of 40 hours per week. FICA taxes are...
-
Determine the force in each member of the truss and state if the members are in tension or compression. Set P 1 = 200 lb, P 2 = 500 lb. 4 ft A -3 ft- B P - P -4 ft-
-
Two of the three wires in Figure P28.8 carry current \(I\), and the third wire carries current 6I. The location labeled \(\mathrm{P}\) is a distance \(d\) from cach of the wires carrying current...
-
Curtis Rich, the cost accountant for Hi-Power Mower Company, recently installed activity-based costing at Hi-Powers St. Louis lawn tractor (riding mower) plant where three modelsthe 8-horsepower...
-
1. Let p and q be two positive numbers such that p + q = 2 and p + q = 272. Then p and q are roots of the equation: (a) x-2x+8=0 2. lim x 0 (sint)dt (b) x-2x+136=0 (c) x-2x+16=0 (d) x-2x+2=0 0
-
How many atoms of silver equal a mass of 107.87 amu? (a) 1 (b) 47 (c) 107.87 (d) 108 (e) 6.02 x 10 23 .
-
Chlorine is prepared industrially by heating hydrogen chloride gas with oxygen gas. Assuming the only products are water and chlorine gas, write a balanced chemical equation for the manufacture of...
-
What is the clonal selection model as it applies to B cells? What becomes of the clones that are produced?
-
Assume Shehata Coffee Shop has a Prepaid Rent account with a beginning balance of $2,000. An additional $15,000 was pre-paid during the year with a debit to Prepaid and credit to Cash. If the ending...
-
Suppose you deposit the cash flows in a bank account that pays 7 % interest per year. What is the balance in the account at the end of each of the next three years (after your deposit of$190 is made)
-
According to Blythe (2013, p. 103), 'perception is a process of converting sensory information into an understanding of how the world works'. It can also be described as a process by which sensory...
-
Consider the pattern 1,4,9,16,25,36,dots. Each number in the pattern is changed to make a new number pattern, which is -2,1,6,13,22,dots. What are the next three terms in the new pattern?
-
An online vacation rental company 'VacationforYou' allows customers to book rentals on their website. Customers can book many vacation rentals and a rental can be booked by one customer at a time....
-
What does the term sales life cycle mean? What are the phases of the sales life cycle? How does it differ from the cost life cycle?
-
Find the image of x = k = const under w = 1/z. Use formulas similar to those in Example 1. y| y = 0 -21 -2 -1 -1, /1 12 T -1 -1 y= -2 x =0
-
Refer to the accounting change by Wertz Construction Company in BE22-1. Wertz has a profit-sharing plan, which pays all employees a bonus at year-end based on 1% of pre-tax income. Compute the...
-
Shannon, Inc., changed from the LIFO cost flow assumption to the FIFO cost flow assumption in 2010. The increase in the prior years income before taxes is $1,200,000. The tax rate is 40%. Prepare...
-
Tedesco Company changed depreciation methods in 2010 from double-declining-balance to straight-line. depreciation prior to 2010 under double-declining-balance was $90,000, whereas straight line...
-
A car traveling 87.0 km/h is 1500 m behind a truck traveling at 74.0 km/h. How far from its initial position does the car have to travel to catch up to the truck.
-
1) For the system in the diagram m2=2.0 kg m3=3.0 kg 0=30 k=0.10 s=0.12. Find m1 and its tension for the following m3 m2 72 a) m1 accelerates upward with a=1.2 m/s^2 m1 m1= T= b) m1 moves upward with...
-
What is the net electric charge of this object? Note for advanced students: you can assume charge is measured in any convenient units, e.g. coulombs or units of e. Write down only the number part of...

Study smarter with the SolutionInn App


