Refer to the periodic table and state the number of atoms in each of the following masses.
Question:
Refer to the periodic table and state the number of atoms in each of the following masses.
(a) 195.08 amu Pt
(b) 195.08 g Pt.
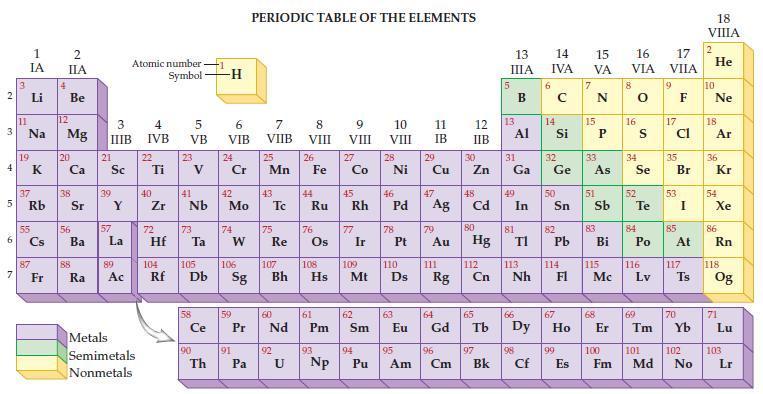
Transcribed Image Text:
2 3 4 AD 6 3 7 11 1 IA 37 5 Rb Li Na 19 55 87 Fr 4 2 IIA Be 12 Mg 20 38 56 Ba 88 Ra 3 IIIB 21 Sc 39 57 Y La 89 Atomic number Symbol Ac Metals Semimetals Nonmetals 4 IVB 22 Ti 40 Zr 5 VB 72 23 41 Nb 73 Hf Ta 104 105 Rf Db Ce 90 Th -H 6 VIB 24 Cr 42 Mo 74 W 106 Sg 59 91 PERIODIC TABLE OF THE ELEMENTS Pa 7 VIIB 25 Mn 43 75 Re 60 Pr Nd 107 Bh 92 U 8 VIII 26 44 Fe Ru 76 108 Hs 61 Pm 93 Np 9 VIII 27 Co 77 Ir 109 62 45 46 47 Rh Pd Ag Sm 10 VIII 110 Mt Ds 94 28 Pu Ni 78 63 11 IB 29 95 Cu 79 Au 111 64 Eu Gd Rg 96 Am Cm 12 13 IIB 30 Zn 48 Cd 80 Hg 112 Cn 65 97 13 IIIA Bk 31 Al Ga 49 In 81 TI 113 Nh 66 98 Cf 14 15 16 IVA VA VIA 6 14 32 Ge 50 Sn 82 Pb 114 67 99 E Es 33 51 34 As Se 83 Bi 115 Mc 68 8 16 52 S Te 84 Po 116 Lv 69 17 VIIA 9 17 cl 35 Br 53 85 At 117 Ts 70 100 101 102 Fm Md No 18 VIIIA 2 He 10 Ne 18 Ar 36 54 Kr Xe 86 Rn 118 Og 71 Lu 103 Lr
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
a One platinum atom ...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Introductory Chemistry Concepts And Critical Thinking
ISBN: 9780321804907
7th Edition
Authors: Charles Corwin
Question Posted:
Students also viewed these Sciences questions
-
Refer to the periodic table and state the number of atoms for each of the following nonmetals. (a) 30.97 g P (b) 20.18 g Ne. Periodic Table: 19 2 3 4 5 6 7 3 11 1 IA Li 6.94 Na 22.99 19 37 4 12 87 2...
-
Refer to the periodic table and state the mass of Avogadros number of atoms for each of the following nonmetals. (a) Sulfur (b) Helium. Periodic Table: 19 2 3 4 5 6 7 3 11 1 IA Li 6.94 Na 22.99 19 37...
-
Refer to the periodic table and state the number of valence electrons for any element in each of the following groups: (a) Group IIA (b) Group VA (c) Group 14 (d) Group 17. Periodic Table: 2 3 4 15...
-
In Exercises, use the alternative form of the derivative to find the derivative at x = c (if it exists). f(x) = 1 x + 4' c = 3
-
Presented below are two independent transactions. Both transactions have commercial substance. 1. Rangi Co. exchanged old trucks (cost $64,000 less $22,000 accumulated depreciation) plus cash of...
-
Use substitution to find each indefinite integral. (-41 + (-4t + 1) dt
-
Planes-for-Rent, Inc., (PFR) leases airplanes. Dayton, LLC, is one of PFRs customers. Dayton is a limited liability company with one member, Jacob, who is Daytons sole owner. Dayton and PFR executed...
-
Sawyer Pharmaceuticals manufactures an over-the-counter allergy medication. The company sells both large commercial containers of 1,000 capsules to health-care facilities and travel packs of 20...
-
after determing a reported vulnerability was a credible claim the product security incident response team worked with develpment teams to create and test a patch. the patch is schedule t obe released...
-
Write a balanced equation for each of the following combination reactions. (a) Sulfur is heated with oxygen to form sulfur dioxide gas. (b) Sulfur is heated with oxygen and Pt catalyst to form sulfur...
-
Refer to the periodic table and state the atomic mass (in amu) of one atom for each of the following nonmetals. (a) Beryllium (b) Barium (c) Boron (d) Bromine. 2 3 4 AD 6 3 7 11 1 IA 37 5 Rb Li Na 19...
-
Write a paper on Human Rights in any Area of the World, Globalization and its impact on any area of the world, Women Rights in the Middle East, Role of the UN in any part of the World (Successful or...
-
Chelsea has saved $20,000 for her son's education to date. He is 10 years old and she has 8 more years to save. If she invests $1,000 a year for the next 8 years, at the end of each year, how much...
-
Multiply the radicals and simplify. 122710
-
Solve. x+3x-22x-7 The solution set is
-
Solve the equation. 2y 8y y-6 y-6
-
An organization is releasing a major application within the next 60 days. The organization asked an auditor to do an assessment. The auditor has insufficient time to review all of the controls but is...
-
Award Plus Co. manufactures medals for winners of athletic events and other contests. Its manufacturing plant has the capacity to produce 10,000 medals each month; current monthly production is 7,500...
-
A Bloomberg Businessweek subscriber study asked, In the past 12 months, when traveling for business, what type of airline ticket did you purchase most often? A second question asked if the type of...
-
How do you think you would like telecommuting?
-
In which stage of development is each of the four groups listed in question 3? Did any group move too quickly through any of the stages? Explain.
-
Among the various approaches to enhancing workplace satisfaction and productivity discussed in the chapter, which ones might you take under the circumstances described in the case? Why are some of...
-
I hired a carpenter and an apprentice to do some work at my business. They worked two days. On the first day I was billed $210 for 11 hours of work by the main carpenter and for 7 hours of work by...
-
It is March, and the annual NCAA Basketball Tournament is down to the final 4 teams. Randy Kitchell is a t-shirt vendor who plans to order t-shirts with the names of the final 4 teams from a...
-
L = {w| L2 = {w| Two languages L1 and L2 are defined by symbols = {0, 1}, written as w contains 00 as a sub-string and the length of w is even number} w begins with 0 and ends with 1} (i) Write two...

Study smarter with the SolutionInn App


