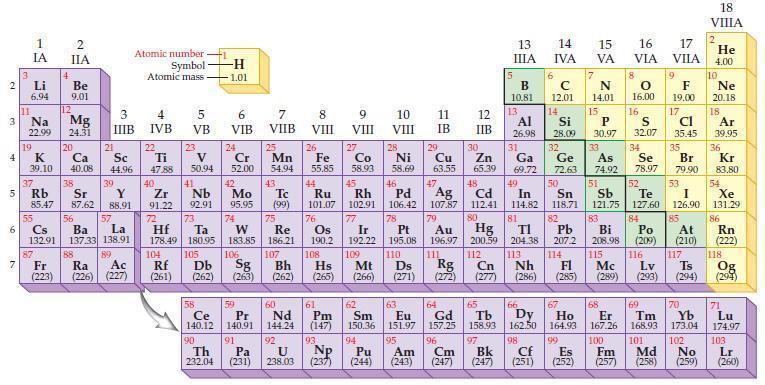
Refer to the periodic table and state the number of valence electrons for any element in each
Question:
Refer to the periodic table and state the number of valence electrons for any element in each of the following groups:
(a) Group IIA
(b) Group VA
(c) Group 14
(d) Group 17.
Periodic Table:
Transcribed Image Text:
2 3 4 15 6 7 3 11 Li 6.94 Na 22.99 19 1 IA 37 R 55 4 12 87 2 IIA Be 9.01 Mg 24.31 K Ca Sc 39.10 40.08 44.96 20 38 21 Rb Sr Y 85.47 87.62 88.91 3 IIIB 88 39 57 56 Cs La Ba 132.91 137.33 138.91 89 Fr Ra Ac (223) (226) (227) Atomic number Symbol Atomic mass 4 IVB 22 Ti 47.88 40 Zr 2 91.22 72 5 VB 104 23 V 50.94 41 73 Hf Ta 178.49 180.95 105 Rf Db (261) (262) -H -1.01 6 VIB Nb Mo 92.91 95.95 90 24 Cr 52.00 42 74 106 Sg (263) 58 Pr Ce 140.12 140.91 91 59 7 VIIB Th Pa 232.04 (231) 25 W Re 183.85 186.21 Mn 54.94 43. Tc (99) 75 107 Bh (262) 60 Nd 144.24 92 U 238.03 8 VIII 26 Fe 55.85 76 Os 190.2 61 Pm (147) 93 NP 9 VIII (237) 27 10 VIII 11 IB 12 IIB 28 29 Co Zn Ni Cu 58.93 58.69 63.55 65.39 44 47 48 49 45 46 Ru Rh Pd Ag Cd In 101.07 102.91 106.42 107.87 112.41 114.82 77 78 80 79 Au Hg 196.97 200.59 111 112 Ir Pt 192.22 195.08 109 110 Hs Mt Ds Rg Cn (265) (266) (271) (272) (277) 108 30 5 13 ΠΙΑ B 10.81 13 6 14 IVA с 12.01 14 81 82 TI Pb 204.38 207.2 113 114 Nh Fl (286) (285) 15 16 17 VA VIA VILA 7 N 14.01 15 66 67 62 63 64 65 Sm Eu Gd Tb Dy Ho 150.36 151.97 157.25 158.93 162.50 164.93 99 94 95 96 97 Pu Am Cm Bk (244) (243) (247) (247) 8 P 30.97 Al Si 31 34 26.98 28.09 32 33 Ga Ge As Se 69.72 72.63 74.92 78.97 50 51 52 Sn Sb Te I 118.71 121.75 127.60 126.90 53 83 84 85 Bi Po At 208.98 (209) (210) 115 116 117 Lv Ts (289) (293) (294) Mc O 16.00 16 9 F 19.00 17 Cl 35.45 S 32.07 35 Br 79.90 68 69 70 Er Tm Yb 167.26 168.93 173.04 100 101 102 Cf Es Fm Md No (251) (252) (257) 98 (258) (259) 18 VIIIA 2 He 4.00 10 Ne 20.18 18 Ar 39,95 36 Kr 83.80 54 Xe 131.29 86 Rn (222) 118 Og (294) 71 Lu 174.97 103 Lr (260)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
a 2...View the full answer

Answered By

Morgan Njeri
Very Versatile especially in expressing Ideas in writings.
Passionate on my technical knowledge delivery.
Able to multitask and able to perform under pressure by handling multiple challenges that require time sensitive solution.
Writting articles and video editing.
Revise written materials to meet personal standards and satisfy clients demand.
Help Online Students with their course work.
4.90+
12+ Reviews
38+ Question Solved
Related Book For 

Introductory Chemistry Concepts And Critical Thinking
ISBN: 9780321804907
7th Edition
Authors: Charles Corwin
Question Posted:
Students also viewed these Sciences questions
-
Refer to the periodic table and predict the number of valence electrons for an atom of each of the following representative elements: (a) Na (b) Al (c) S (d) Xe. Periodic Table: 2 3 4 5 N.. 3 7 Li...
-
Element 43 is used in medical radiology to locate tumors. Refer to the periodic table and state whether Tc has any stable isotopes. Periodic Table: 2 3 4 5 6 7 3 11 1 IA Li Na 19 37 Rb 55 87 Fr 4 2...
-
Element 61 was named for the mythological Greek Prometheus who stole fire from the gods. Refer to the periodic table and state whether Pm has any stable isotopes. Periodic Table: 2 3 4 5 6 7 3 11 1...
-
Explain how you solve problems during the implementation of your projects. Does this operate as a formal approach and/or use specific supportive techniques?
-
Explain the importance of diagnosis-related groups (DRGs) in the cost accounting systems of a health care provider.
-
For each of the following independent situations, determine the appropriate accounting method to be used: cost or equity. For cost method situations, determine whether the security should be...
-
Select a public company that has reported a goodwill impairment in its 10-K report. Possible companies to consider are Baker Hughes Co., A&T Inc., Uber Technologies, Carnival Corporation, Tapestries...
-
Lock Tight, Inc., produces outside doors for installation on homes. The following information was gathered to prepare budgets for the upcoming year beginning January 1: Sales forecast in units . . ....
-
1. The endpoints of the bottom side of a parallelogram are at A(2, 1) and B(10, 7). The other two sides both have slope 3, and length 5 cm. (a) (2 points) Provide a very neat and accurate sketch this...
-
Which two elements in the fourth period violate the original periodic law as stated by Mendeleev?
-
What is the electron dot formula for an atom of neon? (a) (b) (c) (d) (e) Ne
-
On October 31, 2021, Lisik Company had a cash balance per books of $8,946. The bank statement on that date showed a balance of $10,155. A comparison of the statement with the Cash account revealed...
-
Everything is the same as in Critical Discussion Question 3, except the counterfeit products involved are the more affordable generic drugs to combat HIV/AIDS. Providing these drugs at a lower cost...
-
Use the information given in question 7 to find P(X 8). Question 7 Suppose a random variable X can take on only values in the range from 2 to 10 and that the probability that the variable will...
-
On March 23, 2010, President Obama signed into law a major overhaul of the U.S. healthcare system. The Congressional Budget Office estimated that this legislation will reduce the U.S. government...
-
Go to the St. Louis Federal Reserve FRED database, and calculate the GDP growth rate and inflation rate. For GDP, use (GDPC1). For the inflation rate, use the personal consumption expenditure price...
-
Again, use the information given in question 18. This time assume that 100 homes are randomly sampled instead of Question 18 A real estate agent in Connecticut is interested in the mean home price in...
-
Study the following Excel multiple regression output. How many predictors are in this model? How many observations? What is the equation of the regression line? Discuss the strength of the model in...
-
The graph of the sequence whose general term is an = n - 1 is which of the following? [8.1] A. B. TITTT 3-2-1 23.45 2.3.4
-
Unlike the other major financial statements, the statement of cash flows is not prepared from the adjusted trial balance. From what sources does the information to prepare this statement come, and...
-
Why is it necessary to convert accrual-based net income to a cash basis when preparing a statement of cash flows?
-
Differentiate between the direct method and the indirect method by discussing each method.
-
Carlisle Transport had $4,499 cash at the beginning of the period. During the period, the firm collected $1,750 in receivables, paid $2,154 to supplier, had credit sales of $5,578, and incurred cash...
-
Mavericks Cosmetics buys $4,691,301 of product (net of discounts) on terms of 7/10, net 60, and it currently pays on the 10th day and takes discounts. Mavericks plans to expand, and this will require...
-
Mahrouq Technologies buys $19,290,327 of materials (net of discounts) on terms of 3/30, net 60, and it currently pays within 30 days and takes discounts. Mahrouq plans to expand, and this will...

Study smarter with the SolutionInn App


