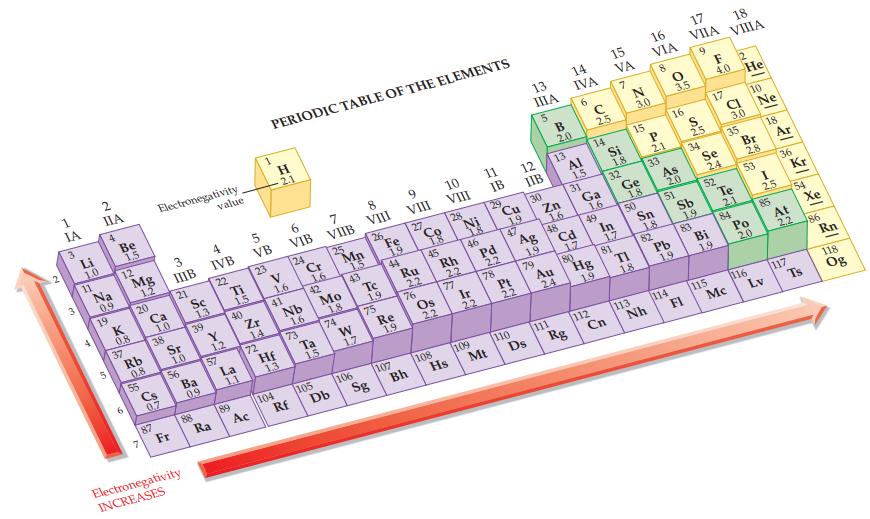
Refer to the values in Figure 12.9 and calculate the electronegativity difference in each of the following
Question:
Refer to the values in Figure 12.9 and calculate the electronegativity difference in each of the following bonds.
(a) H—Cl
(b) H—Br
(c) N—O
(d) C—O.
Figure 12.9
Transcribed Image Text:
IA 3 3 Li 1.0 11 4 IIA Bo Na 0.9 19 5 Be 1,5 12 NE K 0.8 37 Mg 1.2 20 Rb 0.8 55 Ca 1.0 Electronegativity 38 Cs 0,7 87 3 IIIB 21 Sr 1.0 56 Fr Sc 1.3 39 Ba 0.9 Electronegativity INCREASES 88 value 4 IVB 22 Y 1.2 57 Ra Ti 15 89 40 La 1.1 5 VB 23 Zr 14 72 Ac PERIODIC TABLE OF THE ELEMENTS H 2.1 V 1.6 41 Hf 13 104 6 VIB 24 Nb 16 73 Rf Cr 1.6 42 Ta 1.5 7 VIIB 105 25 Mo 1.8 74 Db Mn 1.5 43 W 1,7 8 VIII 106 26 Te 19 75 Sg Fe 22 EN ON 19 44 Re 1.9 9 VIII 107 27 Ru 22 76 Bh Co 45 Os 22 10 VIII 28 108 Rh 22 77 Hs 18 46 Ir 22 11 IB 109 29 Pd 2.2 78 Mt Cu 19 47 Pt 22 110 13 ΠΙΑ 5 12 IIB 30 Ag 1.9 79 Ds B 2.0 Zn 13 16 48 Au 111 24 14 IVA 6 Al 1.5 31 Cd 17 Rg 80 с 2.5 14 Ga 1.6 49 112 Hg 19 15 VA 7 Si 1.8 Cn 32 In 1.7 81 Ge 21 1.8. 15 50 7 TI 18. 3.0 113 16 VIA Nh P 2.1 Sn 18 8 33 82 As 20 Pb 1.9 O 3.5 51 114 17 VIIA 9 16 S 25 34 Sb 1.9 FI 83 Se 24 Bi F He 4.0 17 52 115 19 18 VIIIA CI 3.0 35 Te 2.1 Mc 84 Br 10 2.8 53 Po 20 116 Ne 18 I 25 85 3 Lv Ar 36 At Kr 117 54 2.2 Xe Ts 86 Rn 118 Og
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
a HCl Electronegativity difference 316 220096 b HBr ...View the full answer

Answered By

Chiranjib Thakur
I have no tutoring experience yet, but I can share my skills and knowledge gained from my education and work experiences. I have been a CPA since 2012 with 6 years of work experience in internal auditing and 4 years of work experience in accounting at the supervisory level.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Introductory Chemistry Concepts And Critical Thinking
ISBN: 9780321804907
7th Edition
Authors: Charles Corwin
Question Posted:
Students also viewed these Sciences questions
-
Find the kinetic energy of the a-particle emitted in the decay 28 Pu 234U+ a. The atomic masses needed are as follows: 238 Pu 234 U 238 04955 u 234 04095 u Neglect any recoil of the residual nucleus....
-
Refer to the values in Figure 12.9 and calculate the electronegativity difference in a SI bond. Figure 12.9 1 IA S Li 1.0 11 2 IIA 4 Na 0,9 19 Be 1.5 12 0.8 37 Mg 1.2 6 20 Rb 0.8 55 Ca 1.0 38 3...
-
Preparing and interpreting a statement of cash flows using a T-account work sheet. Financial statement data for Dickerson Manufacturing Company for the current year appear in Exhibit 5.29. Additional...
-
How does an expenditure differ from an expense? Identify the funds that report expenditures and those that report expenses.
-
Greater than 0.25 In assume that a randomly selected subject is given a bone density test. Those test scores are normally distributed with a mean of 0 and a standard deviation of 1. In each case,...
-
Draw an \(x y z\) coordinate system with the \(x\) axis pointing horizontally to the right, the \(y\) axis pointing up the page, and the \(z\) axis pointing out of the page. Show a current-carrying...
-
1. To recruit the CEO from the inside seems to work well for Intel. Do you believe this is a sound policy? Why? 2. How can a nontechnically oriented leader like Paul Otellini succeed as CEO in a...
-
QUESTION 3 (20 MKS) a) Obtain the time-independent Schrodinger Wave equation from the time dependent equation. [10 mks] b) Solve the time-independent Schrodinger equation given that,(x) = Aekx [10...
-
Refer to the values in Figure 12.9 and calculate the electronegativity difference in each of the following bonds. (a) BrCl (b) BrF (c) ICl (d) IBr. Figure 12.9 IA 3 3 Li 1.0 11 4 IIA Bo Na 0.9 19 5...
-
Which elements are more electronegative: semimetals or nonmetals?
-
What are Treasury bonds? Describe their key characteristics.
-
Marie wants to buy a Paw Patrol lunch bag for her grandson. She won't spend any more than $15 on the item, so she's been looking on Amazon.ca. She finds two options under that price. Marie is at what...
-
Consider a symmetrical airfoil with 1 m chord in a wind tunnel experiment where free stream velocity is 37 m/s at sea level conditions. Using the flat plate approximation, estimate the viscous drag...
-
When preforming credit risk analysis, try to assess the following: Expected Credit Loss= Chance of Default Loss Given Default Comment on overall assessment of the company's credit risk and chance of...
-
Draft a new customer profitability statement for both alpha hospital and beta hospital using new activity drivers shown below. Work through the following numerical exercise by filling in the template...
-
Why are the liabilities and cash outlays of a property and casualty insurance company more difficult to predict than for a life insurance company?What are the effects of these differences on the P/C...
-
Which of the following exchanges of property are like-kind exchanges? a. Horace trades his personal use auto for another personal use auto. b. Lian trades an office building she rents out for a...
-
The nitrogen atoms in N2 participate in multiple bonding, whereas those in hydrazine, N2H4, do not. (a) Draw Lewis structures for both molecules. (b) What is the hybridization of the nitrogen atoms...
-
Short-term investments for each if the short-term marketable securities given here provide an example of the potential disadvantages the investment has for meeting a corporations cash management...
-
Agency issues It is sometimes argued that excess cash held by a firm can aggravate agency problems and, more generally, reduce incentives for shareholder wealth maximization how would you frame the...
-
Use of excess cash one option a firm usually has with any excess cash is to pay its suppliers more quickly. What are the advantages and disadvantages of this use of excess cash?
-
Imagine you represent your company at a service organization dealing with one of these two issues: Facing Economic Change Engaging Civil Rights Your supervisor has asked you to research information...
-
Ramirez Company installs a computerized manufacturing machine in its factory at the beginning of the year at a cost of $85,400. The machine's useful life is estimated at 20 years, ar 402,000 units of...
-
Joe just decided to retire from the University of Miwsukee atter 3 0 years of service. As he goes through the retirement process, he learns the details of his defined benefit pension UM affers its...

Study smarter with the SolutionInn App


