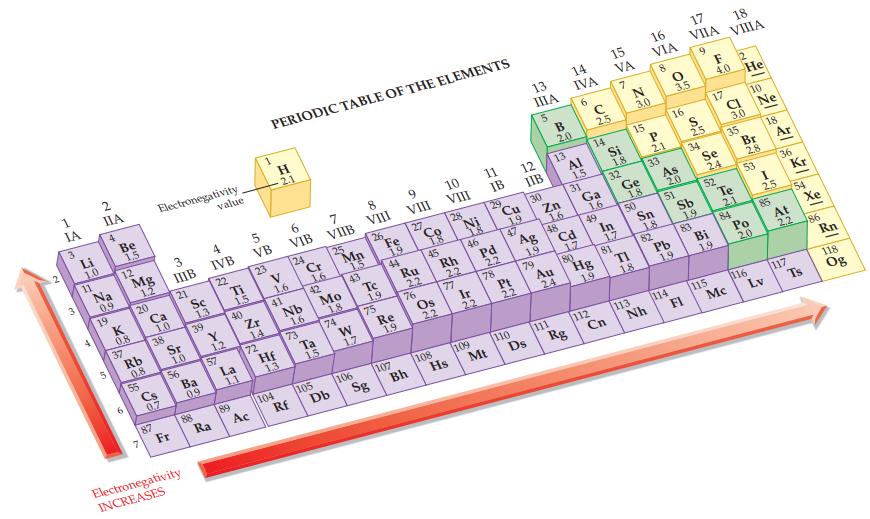
Refer to the values in Figure 12.9 and calculate the electronegativity difference in each of the following
Question:
Refer to the values in Figure 12.9 and calculate the electronegativity difference in each of the following bonds.
(a) Br—Cl
(b) Br—F
(c) I—Cl
(d) I—Br.
Figure 12.9
Transcribed Image Text:
IA 3 3 Li 1.0 11 4 IIA Bo Na 0.9 19 5 Be 1,5 12 NE K 0.8 37 Mg 1.2 20 Rb 0.8 55 Ca 1.0 Electronegativity 38 Cs 0,7 87 3 IIIB 21 Sr 1.0 56 Fr Sc 1.3 39 Ba 0.9 Electronegativity INCREASES 88 value 4 IVB 22 Y 1.2 57 Ra Ti 15 89 40 La 1.1 5 VB 23 Zr 14 72 Ac PERIODIC TABLE OF THE ELEMENTS H 2.1 V 1.6 41 Hf 13 104 6 VIB 24 Nb 16 73 Rf Cr 1.6 42 Ta 1.5 7 VIIB 105 25 Mo 1.8 74 Db Mn 1.5 43 W 1,7 8 VIII 106 26 Te 19 75 Sg Fe 22 EN ON 19 44 Re 1.9 9 VIII 107 27 Ru 22 76 Bh Co 45 Os 22 10 VIII 28 108 Rh 22 77 Hs 18 46 Ir 22 11 IB 109 29 Pd 2.2 78 Mt Cu 19 47 Pt 22 110 13 ΠΙΑ 5 12 IIB 30 Ag 1.9 79 Ds B 2.0 Zn 13 16 48 Au 111 24 14 IVA 6 Al 1.5 31 Cd 17 Rg 80 с 2.5 14 Ga 1.6 49 112 Hg 19 15 VA 7 Si 1.8 Cn 32 In 1.7 81 Ge 21 1.8. 15 50 7 TI 18. 3.0 113 16 VIA Nh P 2.1 Sn 18 8 33 82 As 20 Pb 1.9 O 3.5 51 114 17 VIIA 9 16 S 25 34 Sb 1.9 FI 83 Se 24 Bi F He 4.0 17 52 115 19 18 VIIIA CI 3.0 35 Te 2.1 Mc 84 Br 10 2.8 53 Po 20 116 Ne 18 I 25 85 3 Lv Ar 36 At Kr 117 54 2.2 Xe Ts 86 Rn 118 Og
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
a BrC130 Ax 302...View the full answer

Answered By

Bhartendu Goyal
Professional, Experienced, and Expert tutor who will provide speedy and to-the-point solutions. I have been teaching students for 5 years now in different subjects and it's truly been one of the most rewarding experiences of my life. I have also done one-to-one tutoring with 100+ students and help them achieve great subject knowledge. I have expertise in computer subjects like C++, C, Java, and Python programming and other computer Science related fields. Many of my student's parents message me that your lessons improved their children's grades and this is the best only thing you want as a tea...
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Introductory Chemistry Concepts And Critical Thinking
ISBN: 9780321804907
7th Edition
Authors: Charles Corwin
Question Posted:
Students also viewed these Sciences questions
-
Find the kinetic energy of the a-particle emitted in the decay 28 Pu 234U+ a. The atomic masses needed are as follows: 238 Pu 234 U 238 04955 u 234 04095 u Neglect any recoil of the residual nucleus....
-
Refer to the values in Figure 12.9 and calculate the electronegativity difference in a SI bond. Figure 12.9 1 IA S Li 1.0 11 2 IIA 4 Na 0,9 19 Be 1.5 12 0.8 37 Mg 1.2 6 20 Rb 0.8 55 Ca 1.0 38 3...
-
Refer to the values in Figure 12.9 and calculate the electronegativity difference in a HP bond. Figure 12.9 1 IA S Li 1.0 11 2 IIA 4 Na 0,9 19 Be 1.5 12 0.8 37 Mg 1.2 6 20 Rb 0.8 55 Ca 1.0 38 3...
-
A job order cost accounting system is fully integrated into the general ledger of a company. Identify the major general ledger accounts used in a job order cost system. Explain how manufacturing...
-
What are the three sections of a CAFR? Briefly identify the contents of each section.
-
The U.S. Air Force requires that pilots have heights between 64 in. and 77 in. a. Find the percentage of men meeting the height requirement. b. If the Air Force height requirements are changed to...
-
A wire \(70.0 \mathrm{~mm}\) long is bent in a right angle such that the wire starts at the origin and goes in a straight line to \(x=30.0 \mathrm{~mm}, y=0\), and then in another straight line from...
-
Kellogg Company has its headquarters in Battle Creek, Michigan. The company manufactures and sells ready-to-eat breakfast cereals and convenience foods including cookies, toaster pastries, and cereal...
-
a) Explain the difference between quantum Mechanics and Classical Mechanics b) Explain the Bohr's form of quantization of Energy [4 mks] [3 mks] c) Calculate the penetration distance for a very small...
-
Refer to Figure 12.9 and label each atom in the following polar covalent bonds using delta notation ( + and ). (a) CH (b) SeO (c) PI (d) HBr. Figure 12.9 IA 3 3 Li 1.0 11 4 IIA Bo Na 0.9 19 5 Be...
-
Refer to the values in Figure 12.9 and calculate the electronegativity difference in each of the following bonds. (a) HCl (b) HBr (c) NO (d) CO. Figure 12.9 IA 3 3 Li 1.0 11 4 IIA Bo Na 0.9 19 5 Be...
-
What is a component diagram?
-
Consider the following causal model containing random variables (L, R, B, T}, with dom(T) = {high, low}, dom(B) = {many, few), and all other variables having domain {true, false}. The variable L...
-
The reaction A(aq) + B(aq) Products(aq) was studied, and the following data were obtained: [A]o (mol/L) 0.24 0.12 0.060 0.48 0.36 Submit 0.24 0.18 0.18 [B], (mol/L) The rate constant for the reaction...
-
Dillion Danis Corporation reported net income of $261,360 in 2025 and had 49,000 shares of common stock outstanding throughout the year. Also outstanding all year were 5,200 shares of cumulative...
-
Q1b. Find the sources of the numbers entered in B15, B16, and B17 and list them. 1234 7 600 3 Answers to the questions below will be found by closely examining the case. Detective work is required. 4...
-
Pendant Publishing is considering investing in a new printing press. The initial costs will be $250,000. After-tax cash flows next year will increase by $18,000 and will grow at an annual rate of 4%...
-
Mayfair Corporation exchanges a machine with a fair market value of $15,000 and an adjusted basis of $10,000 for land in Nevada with a fair market value of $15,000. Does Mayfair have a recognized...
-
Write electron configurations for the following ions, and determine which have noble-gas configurations: (a) Cd2+ (b) p3- (c) Zr4+ (d) Ru3+ (e) As3- (f) Ag+
-
Short-tern investments why is referred stock with a dividend tied to short-tern interest rates an attractive short-term investment for corporations with excess cash?
-
Collection and disbursement floats which would a firm prefer: a net collection float or a net disbursement float? Why?
-
Float suppose a firm has a book balance of $ 2 million. At the automatic teller machine (ATM), the cash manager finds out that the bank balance is $2.5 million. What is the situation here? If this is...
-
Nokela Industries purchases a $43.2 million cyclo-converter. The cyclo-converter will be depreciated by $10.8 million per year over four years, starting this year. Suppose Nokela's tax rate is 40%....
-
11.1 Complete the "Rate Category" column on the "Airline" worksheet. After completing the column do exploratory analysis of the airline data to determine how many Airlines fall under Category A? Use...
-
What merge strategy should George and Igman have used to merge the two cultures and why? Possible strategies include assimilation, deculturation, integration or separation.

Study smarter with the SolutionInn App


