Silver metal heated with yellow sulfur powder gives Ag 2 S. Complete the following illustrations by drawing
Question:
Silver metal heated with yellow sulfur powder gives Ag2S.
![]() Complete the following illustrations by drawing the product and excess reactant. Indicate the element that is the limiting reactant in each example.
Complete the following illustrations by drawing the product and excess reactant. Indicate the element that is the limiting reactant in each example.
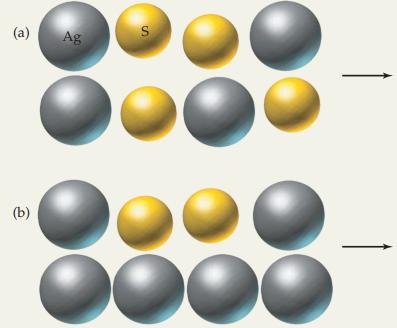
Transcribed Image Text:
A 2 Ag(s) + S(s) Ag₂S(s)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
While its true that heating silver metal with yellow sulfur powder can indeed produce silver sulfide AgS there are some important points to consider C...View the full answer

Answered By

Subash Murugaih
I am leading expert in this web site couple of years and My clients are much happy with my works and services.
4.60+
309+ Reviews
539+ Question Solved
Related Book For 

Introductory Chemistry Concepts And Critical Thinking
ISBN: 9780321804907
7th Edition
Authors: Charles Corwin
Question Posted:
Students also viewed these Sciences questions
-
The Crazy Eddie fraud may appear smaller and gentler than the massive billion-dollar frauds exposed in recent times, such as Bernie Madoffs Ponzi scheme, frauds in the subprime mortgage market, the...
-
The sulfur content of insoluble sulfides that do not readily dissolve in acid can be measured by oxidation with Br2 to SO2-4.25 Metal ions are then replaced with H+ by an ion exchange column, and...
-
The metal is heated in the presence of excess hydrogen, is it obvious which substance is the limiting reagent despite not specifying any quantity of reactant3 Given the statement the metal is heated...
-
A flat and infinitely large sheet with uniform charge density moves with constant speed in a direction parallel to its surface. Confirm the differential form of Poyntings theorem at every point not...
-
Tom Jones says that the balanced scorecard was created to replace financial measures as the primary mechanism for performance evaluation. He says that it uses only nonfinancial measures. Is this true?
-
How do discouraged workers and part-time workers who cannot find full-time jobs alter the true unemployment rate compared to what is officially measured?
-
What benefits should entities expect from preparing sustainability reports?
-
Halo Systems specializes in servers for work- group, e- commerce, and enterprise resource planning (ERP) applications. The companys original job cost system has two direct cost categories: direct...
-
a) Having graduated with a bachelor of commerce finance option, you have been requested to discuss with other employees in finance department on three ways that are used to describe real estate....
-
Classify the following type of stoichiometry problem: How many grams of carbon dioxide gas are produced from the decomposition of 1.10 g NaHCO 3 ?
-
Gasoline and air undergo a combustion reaction in an automobile engine. What is the limiting reactant? What is the excess reactant?
-
Repeat Examples 14.4 and 14.5 on the crime data. Example 14.4. This example compares dCor and Pearson correlation in exploratory data analysis. Consider the Freedman [1975] census data from [United...
-
JaCo uses the periodic method. Its begging inventory is $43,000, purchases are $321,000, FOB destination, purchase returns are $ 17,000,and freight is $ 9,000. The balance in JaCo's ledger Purchases...
-
What senses are included in the 5 cards draw? Describe and explain each of the five senses in complete sentences and how they are used in the event planning process. With two detailed examples in...
-
3. [10 marks] Suppose that the zero-coupon bond prices and oil forward prices for the next four quarters are 1 2 3 4 Oil forward 21 21.1 20.8 20.5 zero-coupon bond 0.9852 0.9701 0.9546 0.9388 (a)...
-
3. (2 points) Predict the major product for the following reaction and provide a stepwise curved arrow mechanism for the formation of the product showing all intermediary products. 1. H H3C C CI AlC...
-
Land Baron Corp. (LBC) owns a parcel of undeveloped land that it acquired in Year 1. Previously, it measured this asset using the cost model. On December 31, Year 4, its year end, the company decided...
-
Choice of Costing System Required The following is a list of Web sites for a number of nonU.S. companies. Briefly describe each company and indicate whether it is more likely to use job costing or...
-
What are the key dimensions of critical thinking 2. Watch the NBC Learn video on Diet Scams. What types of claims are made in this video Are they valid Elaborate on your responses. Discuss this video...
-
As a force for change in the videogame industry, how has technology affected Koticks strategy at Activision? How about competition?
-
In what ways has Robert Kotick acted as a change agent at Activision?
-
Apply Lewins process model of organization change to the measures that Kotick has taken to effect change at Activision. Be sure to examine each step in the Lewin model, noting where the model applies...
-
Using the table below, how much more likely is it that a student from a single - parent household will be eligible for free school meals compared to a student not from a single parent household. You...
-
A partial common-size income statement for Jag Company for three years is shown below. Net sales Item 2019 2018 2017 100.0 100.0 100.0 60.5 63.0 62.5 21.0 19.0 20.5 Cost of goods sold Other expenses...
-
Jakobs, Penn, and Lundt are partners with beginning-of-year capital balances of $400,000, $320,000, and $160,000, respectively. The partners agreed to share income and loss as follows: Salary of...

Study smarter with the SolutionInn App


