State the trends in the periodic table for increasing ionization energy. Periodic Table: 2 3 4 10
Question:
State the trends in the periodic table for increasing ionization energy.
Periodic Table:
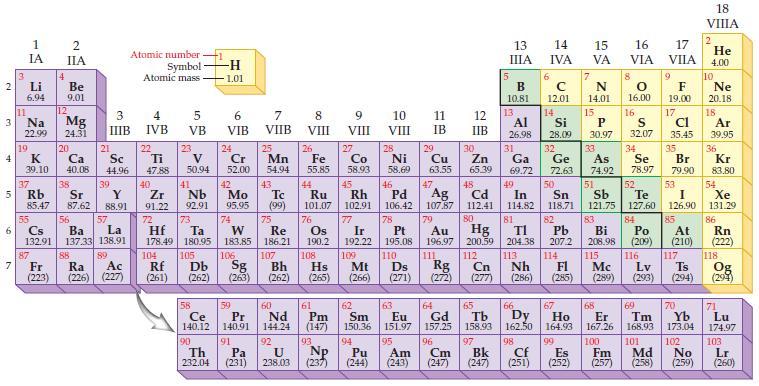
Transcribed Image Text:
2 3 4 10 6 3 7 11 1 IA Li 6.94 Na 22.99 19 37 5 Rb K 39.10 4 87 2 IIA Fr (223) Be 9.01 12 Mg 24.31 20 38 Ca Sc 40.08 44.96 21 Sr Y 85.47 87.62 88.91 55 56 La Cs Ba 132.91 137.33 138.91 88 3 4 IIIB IVB 39 57 89 Atomic number Symbol Atomic mass Ac Ra (226) (227) 22 Ti 47.88 5 VB 23 104 V 50.94 40 Nb Zr 91.22 92.91 41 105 -H 1.01 6 VIB 58 Ce 140.12 90 Th 232.04 24 Cr 52.00 42 Mo 95.95 74 72 73 W Re Hf Ta 178.49 180.95 183.85 186.21 106 Rf Db Sg (261) (262) (263) 59 Pr 140.91 91 7 VIIB Pa (231) 25 Mn 54.94 43 Tc (99) 75 107 60 Nd 144.24 8 VIII 92 26 27 Fe Co 55.85 58.93 Bh Hs (262) (265) 44 45 Ru Rh 101.07 102.91 76 Os 190.2 108 61 Pm (147) 9 VIII 93 U NP 238.03 (237) 77 Ir 192.22 109 Mt (266) 62 Sm 150.36 10 VIII 28 Ni 58.69 46 Pd 106.42 78 11 IB 94 95 Pu Am (244) (243) 13 IIIA 12 IIB 5 6 7 C 12.01 14 N 14.01 15 29 30 31 34 Si P 28.09 30.97 32 33 Cu Zn Ga Ge As Se 63.55 65.39 69.72 72.63 74.92 78.97 48 49 50 52 Ag Cd In Sn Sb Te 107.87 112.41 114.82 118.71 121.75 127.60 80 47 51 B 10.81 13 Al 26.98 14 15 IVA 16 VA VIA 79 81 82 Pt Au Hg TI Pb 195.08 196.97 200.59 204.38 207.2 110 111 112 113 114 Ds Rg Cn Nh Fl (271) (272) (277) (286) 83 Bi 208.98 115 16.00 16 52 S 32.07 84 Po (209) 17 VIIA 9 Md (258) F 19.00 17 Cl 35.45 35 Br 79.90 53 I 126.90 85 At (210) 116 Mc Lv (285) (289) (293) (294) Ts 117 66 70 63 64 65 67 68 69 Eu Gd Tb Dy Но Er Tm Yb 151.97 157.25 158.93 162.50 164.93 167.26 168.93 173.04 96 98 99 100 Cf Es Fm (251) (252) (257) 101 102 Cm (247) 97 Bk (247) 18. VIIIA 2 He 4.00 10 Ne 20.18 18 Ar 39.95 36 Kr 83.80 54 Xe 131.29 86 Rn (222) 118 Og (294) 71 Lu 174.97 103 No Lr (259) (260)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
Ionization energy is increased from left to right across the period because the ...View the full answer

Answered By

Issa Shikuku
I have vast experience of four years in academic and content writing with quality understanding of APA, MLA, Harvard and Chicago formats. I am a dedicated tutor willing to hep prepare outlines, drafts or find sources in every way possible. I strive to make sure my clients follow assignment instructions and meet the rubric criteria by undertaking extensive research to develop perfect drafts and outlines. I do this by ensuring that i am always punctual and deliver quality work.
5.00+
6+ Reviews
13+ Question Solved
Related Book For 

Introductory Chemistry Concepts And Critical Thinking
ISBN: 9780321804907
7th Edition
Authors: Charles Corwin
Question Posted:
Students also viewed these Sciences questions
-
Using the data in Figures 9.15 and 9.16, calculate E for the reaction Na(g) + Cl(g) Na + (g) + Cl (g). Trends in First lonization Energy Ionization energy (kJ/mol) 2500 2000. 1500- 1000- H 1312 0...
-
Refer only to the periodic table on the inside front cover, and arrange the following ionization energies in order of increasing value: the first ionization energy of F; the second ionization energy...
-
The financial information provided below is for two companies which operate in similar retail fields, using the same business and accounting policies A. Calculate for each company, ratios that shows...
-
Let be an arbitrary operation in Problems 5259. Describe the operation for each problem. 503 = 2; 702= 5; 901 = 8; 8 0 2 = 6;
-
OMB Press Release. The following press release from the Office of Management and Budget was issued November 20. 2007. FOR IMMEDIATE RELEASE November 20. 2007 Contact: OMB Communications, 202-395-7254...
-
On June 30, 2011, a flash flood damaged the warehouse and factory of Magna Corporation, completely destroying the work-in-process inventory. There was no damage to either the raw materials or...
-
An outflow of cash would result from which of the following? (a) The decrease in an asset account other than cash. (b) The increase in a liability account. (c) The decrease in a liability account....
-
The following information is from the materials requisitions and time tickets for Job 9- 1005 completed by Great Bay Boats. The requisitions are identified by code numbers starting with the letter Q...
-
sin x 0
-
Predict the common ionic charge for Group VIIA/17 elements; Group VIA/16 elements; Group VA/15 elements.
-
Refer to the periodic table and predict which of the following ions are isoelectronic with the noble gas argon: (a) K + (b) Br (c) Ca 2+ (d) O 2 . 2 3 4 5 6 7 Li 6.94 11 1 IA Na 22.99 19 37 55 4...
-
Your nonprofit club holds a major fund-raiser for two weeks each year to support community improvement projects. The club sells packages of cookies throughout the community and donates the proceeds....
-
Suppose you take an ordinary deck of 52 cards randomly select five cards without replacement. How many different combinations of sample car can you have?
-
The California Public Employees Retirement System (CalPERS) is one of the worlds largest pension funds and one of the earliest to divest stocks of firms that did not meet its ESG criteria. For...
-
Suppose the time you wait at a bank is exponentially distributed with mean 1/ = 12 min. What is the probability that you will wait between 10 and 20 min?
-
Labor economists at the Department of Labor say they have 95 % confidence that factory workers earnings will lie between $22,000 and $61,000. Explain what this means.
-
From past history, a service manager at Honest Abes Auto Dealership knows that 35 % of all customers entering the dealership will have service work done that is under warranty. Suppose 200 people...
-
Use the following data to develop a curvilinear model to predict y. Include both x1and x2 in the model in addition to x21 and x22, and the interaction term x1 x2. Comment on the overall strength of...
-
QUESTION 2 The CEO of Farisha Hijab Sdn Bhd insisted on further investigation to be carried out that he also required Mr Muaz to conduct the analysis of variance for the material and labour of the...
-
Briefly describe some of the similarities and differences between U.S. GAAP and iGAAP with respect to the accounting for leases.
-
Both iGAAP and U.S. GAAP require footnote disclosure of operating lease payments. Are there any differences in the information provided to statement readers in these disclosures? Explain.
-
Briefly discuss the IASB and FASB efforts to converge their accounting guidelines for leases.
-
Standard Costs Labor Materials Total Items per unit 2.00 hrs 1.00 Rate Per Item $ 25.00 $ 70.00 Variable Cost Per Unit Produced $ 50.00 $ 70.00 $ 120.00 During the period, the company produced 1,500...
-
This assignment is an important starting point for the project, so be sure to dedicate some time to doing preliminary research and making the correct choice. To begin, identify a company to use for...
-
The trial balance before adjustments as at 30 June 2023. Can you please help me put together the statement of financial position and the statement of profit and loss and other comprehensive income...

Study smarter with the SolutionInn App


