Sterling silver contains silver and copper metals. If a sterling silver chain contains 18.5 g of silver
Question:
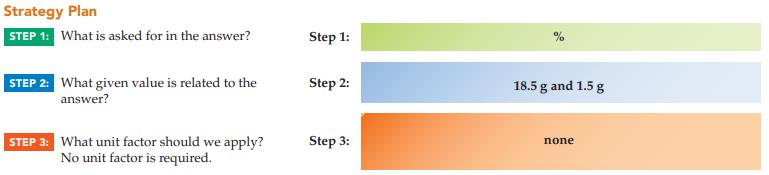
Sterling silver contains silver and copper metals. If a sterling silver chain contains 18.5 g of silver and 1.5 g of copper, what is the percent of silver?
Transcribed Image Text:
◄ Sterling Silver Sterling silver has a high luster and is found in fine utensils and jewelry.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (1 review)
To find percent we compare the mass of silver metal to the total mass o...View the full answer

Answered By

Smita Das
0.00
0 Reviews
10+ Question Solved
Related Book For 

Introductory Chemistry Concepts And Critical Thinking
ISBN: 9780321804907
7th Edition
Authors: Charles Corwin
Question Posted:
Students also viewed these Sciences questions
-
What is the percent yield in Exercise 4? In exercise 4 A worker synthesizes aspirin, C9H8O4, according to this chemical equation. If 12.66 g of C7H6O3 are reacted and 12.03 g of aspirin are isolated,...
-
A tablet PC contains 3217 music files. The distribution of file size is highly skewed with many small file sizes. Assume that the standard deviation for this population is 3.25 megabytes (MB)....
-
The following are the financial statements of Swifty Corporation. Swifty Corporation Comparative Balance Sheets December 31 Assets 2019 2018 Cash $37,200 $19,700 Accounts receivable 33,000 18,400...
-
Problem 4.2 Ask the user to enter his/her age, with the prompt: "How old are you? Please enter your age as a number between 0 and 120. -> ". Check that what was entered is a number between 0 and 120....
-
The function f(x) = 2e-1.5x can be used to generate the following table of unequally spaced data: Evaluate the integral from = 0 to b = 0.6 using (a) Analytical means, (b) The trapezoidal rule, and...
-
Happy Snacks Ltd. (HSL) is a distributor of snack products to various retail stores in Toronto. HSL establishes agreements with stores whereby it places racks in the stores that display HSL's...
-
For approximately 20 months, Robert E. McDonald perpetrated a scheme to solicit millions of dollars purportedly for a \($100\) million purchase by the RAI Entities and certain other related corporate...
-
As the production planner for Scott Sampson Products, Inc., you have been given a bill of material for a bracket that is made up of a base, two springs, and four clamps. The base is assembled from...
-
Mary, Shannon, and Anika each invested $9,000, $25,000, and $18,000, respectively, into a business. a. Calculate their investment ratio reduced to the lowest terms. 0 : 0 : 0
-
If 50.0 mL of gasohol has a mass of 37.5 g, what is the density of the gasohol in grams per cubic centimeter? (a) 0.00750 g/cm 3 (b) 0.0750 g/cm 3 (c) 0.750 g/cm 3 (d) 7.50 g/cm 3 (e) 10.0 g/cm 3 .
-
State the quantity that is measured using the following instruments. (a) Micrometer (b) Electronic balance (c) Buret (d) Atomic clock.
-
Discuss the success factors of BPR in detail.
-
A sales report generated by software run by a small rural caf, 'Beautiful Outlier' was taken to analyse dairy-free milk demand by its customers (and therefore allow the caf owner to make more...
-
All Your discussion should be argumentative question 1.Discuss future consumer purchase behavior trends in the hospitality industry. 2.Discuss how artificial intelligence (AI) have greater...
-
What does the "DRY" principle advocate for in software design?
-
An introduction with a detailed description of the company SWOT, A SWOT analysis diagram that includes strengths, weaknesses, opportunities, and threats An evaluation of how specific internal factors...
-
Describe what Lean operations mean (with a supporting basis). Explain how Lean techniques could be applied to and benefit an area of operations (i.e. forecasting, product development, supply change...
-
The Waterworks Department of the City of Pleasantville provides services to both the Parks and Recreation Department and the citizens of Pleasantville. Recently, a citizen has approached you to...
-
Sandcastles, Inc.s management has recently been looking at a proposal to purchase a new brick molding machine. With the new machine, the company would not have to buy bricks. The estimated useful...
-
A 30-year mortgage for $145,000 is issued at a 6% A nominal interest rate. (a) What is the monthly payment? (b) How long does it take to payoff the mortgage, if $1000 per month is paid? (c) How long...
-
Solve Problem 6-32 for the breakeven first cost per kilometer of going under the lake.
-
Redo Problem 6-38 to calculate the EUAW of the alternatives as a function of miles driven per year to see if there is a crossover point in the decision process. Graph your results.
-
Your analysis shows that the mean capacitance of a batch of 500 of the capacitors you have selected is 46 F, with a standard deviation of 4 F. Assuming the capacitors are normally distributed,...
-
A 0.20-kg block moves at the end of a 0.50 m string along a circular path on a friction-less air table. The block's initial rotational speed is 2.0 rad/s. As the block moves in the circle, the string...
-
Using two-way Set-Associative mapping method, design two-way mapping for cache memory from main memory. Explain why you need each condition and show all intermediate calculation procedures. You must...

Study smarter with the SolutionInn App


