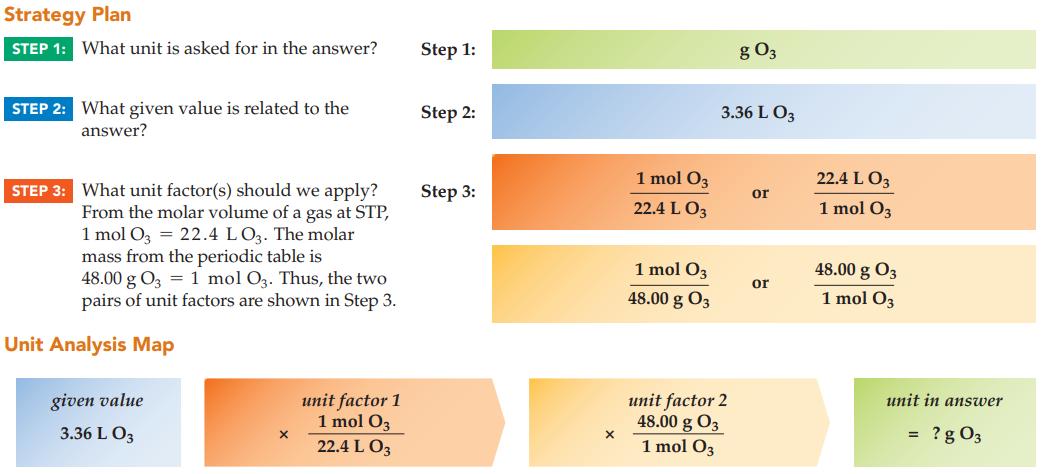
What is the mass of 3.36 L of ozone gas, O 3 , at STP? Strategy Plan
Question:
What is the mass of 3.36 L of ozone gas, O3, at STP?
Transcribed Image Text:
Strategy Plan STEP 1: What unit is asked for in the answer? STEP 2: What given value is related to the answer? STEP 3: What unit factor(s) should we apply? From the molar volume of a gas at STP, 1 mol O3 = 22.4 LO3. The molar mass from the periodic table is 48.00 g 03 1 mol O3. Thus, the two pairs of unit factors are shown in Step 3. Unit Analysis Map given value 3.36 L 03 X unit factor 1 1 mol 03 22.4 L 03 Step 1: Step 2: Step 3: X 1 mol 03 22.4 L 03 1 mol 03 48.00 g 03 g 03 3.36 L 03 unit factor 2 48.00 g 03 1 mol O3 or or 22.4 L 03 1 mol 03 48.00 g 03 1 mol O3 unit in answer ? g 03 =
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (4 reviews)
We apply the unit factor 1 mol 0...View the full answer

Answered By

PALASH JHANWAR
I am a Chartered Accountant with AIR 45 in CA - IPCC. I am a Merit Holder ( B.Com ). The following is my educational details.
PLEASE ACCESS MY RESUME FROM THE FOLLOWING LINK: https://drive.google.com/file/d/1hYR1uch-ff6MRC_cDB07K6VqY9kQ3SFL/view?usp=sharing
3.80+
3+ Reviews
10+ Question Solved
Related Book For 

Introductory Chemistry Concepts And Critical Thinking
ISBN: 9780321804907
7th Edition
Authors: Charles Corwin
Question Posted:
Students also viewed these Sciences questions
-
What are some of the characteristics of the Canadian state that have influenced the way media have developed in Canada? What are some of the ways in which the federal government has played a central...
-
What is the mass of the solid NH4Cl formed when 73.0 g of NH3 are mixed with an equal mass of HCl? What is the volume of the gas remaining, measured at 14.0C and 752 mmHg? What gas is it?
-
What is the mass of solute in 3.81 L of 0.0232 M Zn(NO3)2?
-
An employee earns $24 per hour and 1.5 times that rate for all hours in excess of 40 hours per week. Assume that the employee worked 43 hours during the week. Assume that the FICA tax rate is 7.5%...
-
Presented below are three independent situations. 1. Wakarusa Corporation retired $130,000 face value, 12% bonds on June 30, 2012, at 102. The carrying value of the bonds at the redemption date was...
-
Zoe and William Hawkins are in their mid-30s and have two children, ages 8 and 5. They have combined annual income of $95,000 and own a house in joint tenancy with a market value of $410,000, on...
-
Two large, flat current-carrying sheets are placed parallel to each other, one sheet above the other. The upper sheet carries a current density of \(2.0 \mathrm{~A} / \mathrm{m}\) to the left, and...
-
The Barrett Textile Mill was checked by inspectors enforcing Occupational Safety and Health Administration (OSHA) codes. The inspectors found violations in four categories: hazardous materials, fire...
-
12345 1 2 3 4 5 6 7 8 819 9 10 Define production system and explain types of productions system? Distinguish between product layout and process layout? Explain the principles of good plant layout?...
-
Given that I-127 is the only natural isotope of iodine, what is the mass of one I atom and Avogadros number of I atoms, respectively?
-
What is the volume of one mole of any gas at STP?
-
If f(x) = x 3 , compute f(-5) and f(-5).
-
Explain how the monthly wage tax form works. How do I come up with the calculations?
-
According to Psychology of Waiting Lines, pre-process waits are perceived longer than in-process waits. Explain.
-
How to include an argument/parameter variable to this existing code? # Constant variables assignmentweight= 0.3 quizweight= 0.3 examweight= 0.4 def calculateGPA(): #Input the student's name...
-
A startup has 1,000 shares outstanding. A VC offers to invest $300,000 at a pre-money price of $1,500 per share. What is the pre-money valuation? Give your answer in millions of dollars (for example,...
-
If you were about to receive a 30 year fixed interest mortgage for $280,000 at 3.3% interest, what would your monthly payments be?
-
Explain what is meant by the term management by exception. What is the relationship between the process of standard cost variance analysis and management by exception?
-
Calculate I, , and a for a 0.0175 m solution of Na 3 PO 4 at 298 K. Assume complete dissociation. How confident are you that your calculated results will agree with experimental results?
-
Lower-of-Cost-or-Market Remmers Company manufactures desks. Most of the company's desks are standard models and are sold on the basis of catalog prices. At December 31, 2010, the following finished...
-
Lower-of-Cost-or-Market Garcia Home Improvement Company installs replacement siding, windows, and louvered glass doors for single family homes and condominium complexes in northern New Jersey and...
-
Entries for Lower-of-Cost-or-Market'Direct and Allowance Malone Company determined its ending inventory at cost and at lower-of-cost-or-market at December 31, 2009, December 31, 2010, and December...
-
Shelby Woods owns and operates an ice cream factory, the finest of its kind in all the world. Given that pints of ice cream are meant to be identical (for the same flavor), her company uses process...
-
A manufacturing company reports the following information. Raw materials inventory, ending Raw materials used Current Year $ 169,500 2,160,000 1 Year Ago 2 Years Ago $ 190,500 2,522,000 $ 197,500...
-
Discuss the effects of all five major accounting assumptions on the accounting process

Study smarter with the SolutionInn App


