Question: Without referring to Table 11.6, predict which compound in each of the following pairs has the higher heat of fusion (cal/mol): (a) H 2 O
Without referring to Table 11.6, predict which compound in each of the following pairs has the higher heat of fusion (cal/mol):
(a) H2O or H2S
(b) H2S or H2Se.
Table 11.6
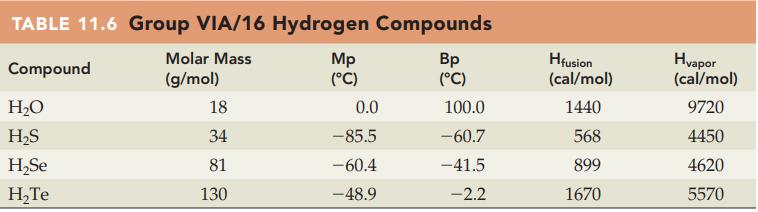
TABLE 11.6 Group VIA/16 Hydrogen Compounds Molar Mass Compound (g/mol) HO HS HSe HTe 18 34 81 130 Mp (C) 0.0 -85.5 -60.4 -48.9 Bp (C) 100.0 -60.7 -41.5 -2.2 Hfusion (cal/mol) 1440 568 899 1670 Hvapor (cal/mol) 9720 4450 4620 5570
Step by Step Solution
3.49 Rating (152 Votes )
There are 3 Steps involved in it
Predicting Heat of Fusion Heres how we can predict the compound with the higher heat of fusion in ea... View full answer

Get step-by-step solutions from verified subject matter experts


