Derive the equations of state (3.52) for an electron-degenerate hydrogen gas. EF = = m cosh(t/4),
Question:
Derive the equations of state (3.52) for an electron-degenerate hydrogen gas.
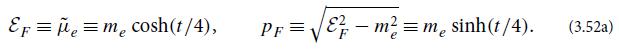
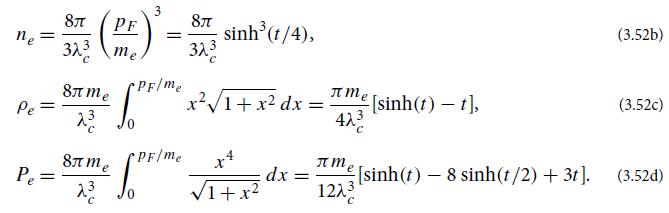
Transcribed Image Text:
EF = μ = m₂ cosh(t/4), PF= |C –mẻ=mesinh(t/4). (3.52a)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 57% (7 reviews)
To derive the equations of state for an electrondegenerate hydrogen gas we need ...View the full answer

Related Book For 

Modern Classical Physics Optics Fluids Plasmas Elasticity Relativity And Statistical Physics
ISBN: 9780691159027
1st Edition
Authors: Kip S. Thorne, Roger D. Blandford
Question Posted:
Students also viewed these Physics questions
-
Derive the equations of the deflection curve for a simple beam AB loaded by a couple M0 acting at distance a from the left-hand support (see figure). Also, determine the deflection δ0 at...
-
Derive the equations of the deflection curve for a cantilever beam AB carrying a uniform load of intensity q over part of the span (see figure). Also, determine the deflection δB at the...
-
Derive the equations of the deflection curve for a cantilever beam AB supporting a distributed load of peak intensity q0 acting over one-half of the length (see figure). Also, obtain formulas for the...
-
Find the point-slope form of the line pass- ing through the given points. Use the first point as (x 1 , y 1 ). (-3,4), (2,5)
-
What are the two types of maturity-related financial markets?
-
Optima Company is a high-technology organization that produces a mass-storage system. The design of Optima's system is unique and represents a breakthrough in the industry. The units Optima produces...
-
Cite various Solution Space tools that enable a homeowner to leverage their time, resources, and skills to maintain their lawn.
-
You are a manager for Herman Millera major manufacturer of office furniture. You recently hired an economist to work with engineering and operations experts to estimate the production function for a...
-
The crew of a helicopter need to land temporarily in a forest and spot a flat horizontal piece of ground (a clearing in the forest) as a potential landing site, but are uncertain whether it is wide...
-
You are working as an analyst for a large cable company that offers bundles of channels all across the United States. One of the bundles is the "basic package," which includes network channels along...
-
Canonical transformations are treated in advanced textbooks on mechanics, such as Goldstein, Poole, and Safko or, more concisely, Landau and Lifshitz (1976). This exercise gives a brief introduction....
-
Consider a collection of identical, classical (i.e., with 1) particles with a distribution function N that is thermalized at a temperature T such that k B T mc 2 (nonrelativistic temperature).(a)...
-
Suppose that the random variable X has the following p.d.f.: Construct a random variable Y = r(X) that has the uniform distribution on the interval [0, 5]. 2e-2 for> o, f(x)-| 2e- for x > 0. 0...
-
A change in the volume occupied by a set of distinguishable particles increases the number of basic states by a factor of 6633 and increases the entropy per particle by a factor of 1. 10. How many...
-
Rank the following systems according to their entropy, lowest first: A. 1,000,000 different colors that can be assigned to each of the \(70 \times 10^{21}\) stars in the observable universe B. The...
-
The rate of change of entropy with respect to thermal energy is \(1.81 \times 10^{20}\) in a system at \(400 \mathrm{~K}\). At what absolute temperature is the rate of change twice this value?
-
An ideal monatomic gas is at \(125^{\circ} \mathrm{C}\). If you increase the thermal energy of the gas and the root-mean-square speed of the gas particles triples, by what factor does the absolute...
-
What is the root-mean-square speed of four monatomic ideal gas particles that have the following velocities (written in component form \():(4.0,6.0,2.0) \mathrm{m} / \mathrm{s},(8.0,-3.0,8.0)...
-
One kg of air at 300 K is mixed with one kg air at 400 K in a process at a constant 100 kPa and Q = 0. Find the final T and the entropy generation in the process.
-
The first law of thermodynamics is sometimes whimsically stated as, You cant get something for nothing, and the second law as, You cant even break even. Explain how these statements could be...
-
Zack is driving past his house in FIGURE Q4.10. He wants to toss his physics book out the window and have it land in his driveway. If he lets go of the book exactly as he passes the end of the...
-
In FIGURE Q4.11, Yvette and Zack are driving down the freeway side by side with their windows down. Zack wants to toss his physics book out the window and have it land in Yvettes front seat. Ignoring...
-
In uniform circular motion, which of the following quantities are constant: speed, instantaneous velocity, the tangential component of velocity, the radial component of acceleration, the tangential...
-
Protecting information resources. 1. What are six examples of information technologies that could be used in computer crimes. 2. What is the CIA triangle? 3. What are three most common security...
-
8. Explain the difference between legislation and common law and provide one (1) example of legislation (using the correct naming convention) and one (1) example of a common law principle. The...
-
Supreme Clothing Use bullet point format Create SWOT diagram Current Strategy Business Model Describe how the firm creates value and drives its growth.

Study smarter with the SolutionInn App


