Question: Evaluate (a) 5. (c) -d.x [*20 0 xe-4x dx ofe (e) e-2t cos.x dx (b) (d) (f) 0 1 x + 2x + 2 In
Evaluate
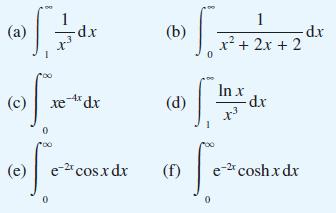
(a) 5. (c) -d.x [*20 0 xe-4x dx ofe (e) e-2t cos.x dx (b) (d) (f) 0 1 x + 2x + 2 In x x3 [e -d.x e-2t cosh xdx d.x
Step by Step Solution
3.40 Rating (159 Votes )
There are 3 Steps involved in it
a b c d e f l... View full answer

Get step-by-step solutions from verified subject matter experts


