The observed dissociation energy of solid LiBr is 788 kJ/mol. Compute the cohesive energy of LiBr and
Question:
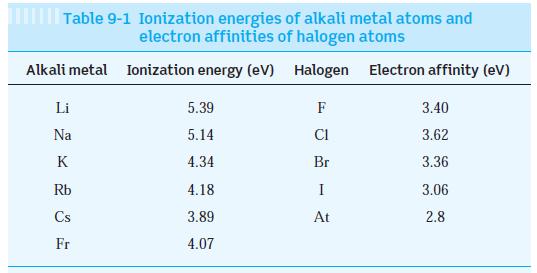
The observed dissociation energy of solid LiBr is 788 kJ/mol. Compute the cohesive energy of LiBr and compare the result with the value in Table 10-1. (Ionization energies for Li and Br are in Table 9-1.)
Table 10-1
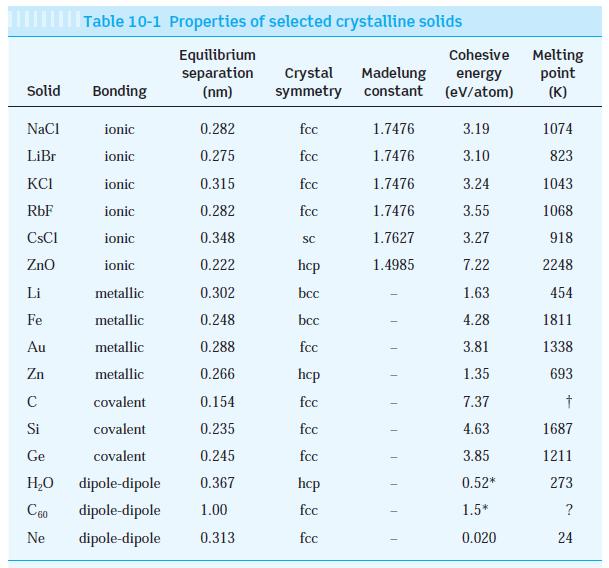
Table 9-1
Transcribed Image Text:
Solid NaCl LiBr KCI RbF ionic 0.282 ionic 0.275 ionic 0.315 ionic 0.282 ionic 0.348 ionic 0.222 metallic 0.302 metallic 0.248 metallic 0.288 metallic 0.266 covalent 0.154 covalent 0.235 Ge covalent 0.245 H₂O dipole-dipole 0.367 C60 dipole-dipole 1.00 Ne dipole-dipole 0.313 CsCl ZnO Table 10-1 Properties of selected crystalline solids Equilibrium separation (nm) Li Fe Au Zn C Si Bonding Crystal Madelung symmetry constant fcc fcc fcc fcc SC hcp bcc bcc fcc hcp fcc fcc fcc hcp fcc fcc 1.7476 1.7476 1.7476 1.7476 1.7627 1.4985 T 1 T Cohesive Melting energy (eV/atom) point (K) 3.19 3.10 3.24 3.55 3.27 7.22 1.63 4.28 3.81 1.35 7.37 4.63 3.85 0.52* 1.5* 0.020 1074 823 1043 1068 918 2248 454 1811 1338 693 † 1687 1211 273 ? 24
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (4 reviews)
Cohesive energy LiBr This is ...View the full answer

Answered By

John Aketch
I have a 10 years tutoring experience and I have helped thousands of students to accomplish their educational endeavors globally. What interests me most is when I see my students being succeeding in their classwork. I am confident that I will bring a great change to thins organization if granted the opportunity. Thanks
5.00+
8+ Reviews
18+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Physics questions
-
The ionization energies of sodium (in kJ/mol), starting with the first and ending with the eleventh, are 495.9, 4560, 6900, 9540, 13,400, 16,600, 20,120, 25,490, 28,930, 141,360, 170,000. Plot the...
-
The successive ionization energies for an unknown element are I1 = 896 kJ/mol I2 = 1752 kJ/mol I3 = 14,807 kJ/mol I4 = 17,948 kJ/mol To which family in the periodic table does the unknown element...
-
The first ionization energies of As and Se are 0.947 MJ/ mol and 0.941 MJ/mol, respectively. Rationalize these values in terms of electron configurations.
-
A 35 ft3 rigid tank has air at 225 psia and ambient 600 R connected by a valve to a piston cylinder. The piston of area 1 ft2 requires 40 psia below it to float, Fig. P3.99. The valve is opened and...
-
For Exercises, write the distribution for the formula and determine whether it is a probability distribution. P(X) = X / (X + 2) for X = 0, 1, 2
-
Flexible work schedules are becoming more common in the workplace, with an increasing number of companies adopting new policies to meet a growing demand for this flexibility from todays labor force....
-
Describe the purpose of an Affirmative Action Plan.
-
The managing directors of Wrack plc are considering what value to place on Trollope plc, a company which they are planning to take over in the near future. Wrack plc's share price is currently 4.21...
-
Crane Co. has zero units of beginning work in process. During the period, 15,120 units were completed and transferred out, and there were 630 units of ending work in process one-fifth complete as to...
-
55 Jason Ready, single, attended the University of San Diego during 2015-2019 under the Army ROTC program. Excluding the school expenses covered by his ROTC scholarship, he incurred additional school...
-
(a) Using = 0.37 nm and (v) = 1.08 x 10 5 m/s at T = 300 K, calculate and for copper from Equations 10-13. Using the same value of , find and at (b) T = 200 K and (c) T = 100 K. me(v) nex and ...
-
The crystal structure of KCl is the same as that of NaCl. (a) Calculate the electrostatic potential energy of attraction of KCl, assuming that r 0 is 0.314 nm. (b) Assuming that n = 9 in Equation...
-
Which document in a job order costing system would show the amount of overtime worked by a specific individual? Explain. Discuss.
-
Evaluate using the improper integral definition. 00 S 3 1 (x-2) dx
-
What is the first step in planning a retrosynthesis of the amine shown below. 1st retrosynthesis step?
-
Would you expect the electron density from nitrogen's lone pair to be closer to the nitrogen in NF3, or in NH3?
-
Let the following data be given: Delta (variation of the price of the option in relation to the price of the underlying) = 0.6; price of the underlying = 100; Value of a call on an underlying...
-
Comment on black swan events by responding to the following questions: What were the causes of these dramatic currency depreciations? What impact could black swan events (and the subsequent...
-
(a) What are some of the crucial considerations that must be taken into account before engaging in an international business project? (b) How does the Foreign Corrupt Practices Act limit the activity...
-
What key concerns must functional tactics address in marketing? Finance? POM? Personnel?
-
Determine the signs (positive, negative, or zero) of the position, velocity, and acceleration for the particle in FIGURE Q1.8. Figure Q1.8
-
FIGURE EX1.9 shows five points of a motion diagram. Use Tactics Box 1.3 to find the average acceleration vectors at points 1, 2, and 3. Draw the completed motion diagram showing velocity vectors and...
-
FIGURE EX1.9 shows five points of a motion diagram. Use Tactics Box 1.3 to find the average acceleration vectors at points 1, 2, and 3. Draw the completed motion diagram showing velocity vectors and...
-
Williams Gear makes and sells three types of computer laptop sleeves: leather, fabric, and plastic. Management is trying to determine the most profitable mix. Sales prices, demand, and use of...
-
Milden Company is a merchandiser that plans to sell 4 1 , 0 0 0 units during the next quarter at a selling price of $ 5 9 per unit. The company also gathered the following cost estimates for the next...
-
How do organizational learning theories, such as the learning organization concept and knowledge management practices, contribute to sustained competitive advantage and organizational resilience in...

Study smarter with the SolutionInn App


