Question: (a) Using = 0.37 nm and (v) = 1.08 x 10 5 m/s at T = 300 K, calculate and for copper
(a) Using λ = 0.37 nm and (v) = 1.08 x 105 m/s at T = 300 K, calculate σ and ρ for copper from Equations 10-13. Using the same value of λ, find σ and ρ at
(b) T = 200 K and
(c) T = 100 K.
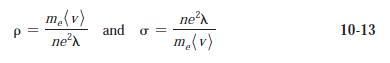
me(v) nex and me(v) 10-13
Step by Step Solution
3.26 Rating (158 Votes )
There are 3 Steps involved in it
a b c p Q m v ne Equation 1... View full answer

Get step-by-step solutions from verified subject matter experts


