Question: Use Equation 10-29 with a = 2 /4 to calculate the average energy of an electron in copper at T = 300 K. Compare
Use Equation 10-29 with a = π2/4 to calculate the average energy of an electron in copper at T = 300 K. Compare your result with the average energy at T = 0 and the classical result of (3/2)kT.
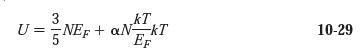
3 kT U = NEF + aNkT EF 10-29
Step by Step Solution
3.45 Rating (152 Votes )
There are 3 Steps involved in it
The average energyelectron at 300K is only 00002eV larger than at 0K a consequence ... View full answer

Get step-by-step solutions from verified subject matter experts


