The ground electronic state of Cr 3+ in an octahedral field is 4 A 2 g ,
Question:
The ground electronic state of Cr3+ in an octahedral field is 4A2g, which derives from a 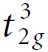 configuration. Is this state subject to Jahn–Teller distortion?
configuration. Is this state subject to Jahn–Teller distortion?
Transcribed Image Text:
3 28
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 62% (8 reviews)
The JahnTeller theorem predicts that a nonlinear molecule or a nonlinear complex ion in a degenerate ...View the full answer

Answered By

Akash M Rathod
I have been utilized by educators and students alike to provide individualized assistance with everything from grammar and vocabulary to complex problem-solving in various academic subjects. I can provide explanations, examples, and practice exercises tailored to each student's individual needs, helping them to grasp difficult concepts and improve their skills.
My tutoring sessions are interactive and engaging, utilizing a variety of tools and resources to keep learners motivated and focused. Whether a student needs help with homework, test preparation, or simply wants to improve their skills in a particular subject area, I am equipped to provide the support and guidance they need to succeed.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The peak in the UV-VIS spectrum of acetone [(CH3)2CO] corresponding to the n * transition appears at 279 nm when hexane is the solvent, but shifts to 262 nm in water. Which is more polar, the ground...
-
The peak in the UV-VIS spectrum of acetone [(CH3)2CoeO] corresponding to the n * transition appears at 279 nm when hexane is the solvent, but shifts to 262 nm in water. Which is more polar, the...
-
Managing Scope Changes Case Study Scope changes on a project can occur regardless of how well the project is planned or executed. Scope changes can be the result of something that was omitted during...
-
EG Corporation redeemed 200 shares of stock from one of its shareholders in exchange for $200,000. The redemption represented 20% of the corporation's outstanding stock. The redemption was treated as...
-
Presented below are data taken from the records of Natural Homes Company. Additional information: 1. Held-to-maturity securities carried at a cost of $25,000 on December 31, 2013, were sold in 2014...
-
Five years ago, your favorite aunt won a $1,000,000 lottery. The prize money is paid out $50,000 per year for twenty years. Unfortunately, your aunt needs as much as $250,000 cash now to pay for...
-
Given that charge separation increases the electric potential energy of a system, what can you conclude about the criteria for keeping a system of positively and negatively charged particles in...
-
Edna Recording Studios, Inc., reported earnings available to common stock of $4,200,000 last year. From those earnings, the company paid a dividend of $1.26 on each of its 1,000,000 common shares...
-
1. Identify products A and B 2. 3. CH3 dil. KMnO4 CrO3 A- B 273 K CH3 CH3 (A) A: OH B: OH OH CH3 CH3 (B) A: OH OH B: (C) A: OHCCH,CH,CH, CCH, B: HOOCCH,CH,CH, CH3 CH3 (D) A: B: OH -CH3 The electrode...
-
The SO 2 molecule, analogous to H 2 O, has a 1 A 1 ground electronic state with the valence electronic configuration (1a 2 ) 2 (4b 2 ) 2 (6a 1 ) 2 (2b 1 ) 0 and low-lying excited singlet states of...
-
The compound shown below has ma x = 736 nm in hexane and 553 nm in water. Explain the basis for the solvent shift. H3C H3C Z NO
-
Reconsider Prob. 8121. Letting the pressure drop vary from 1 kPa to 10 kPa, evaluate the flow rate at intervals of 1 kPa, and plot it against the pressure drop. Data from Problem 8121 A Venturi meter...
-
If it takes a total resource cost of $10,000 to produce 25 computers in the U.S., how many computers would be produced in the U.S. with a total resource cost of $3,200
-
1.Examine the digital market environment and discuss all necessary components for a functional online business. The number will depend on your reading and research. 2. How does a 'situation gap...
-
Returning to the Shipbreakers content, do these people have the choice to work in the shipyards? Or rather, do they have the freedom to leave given the logisticalconstraints of their lives? What does...
-
Enrique must submit his Form 8843 for tax year 2023 by April 15, 2024? a. True b. False submitted
-
You invest $9,000 per year (the sum of your portion and the employer match) into a retirement account starting at age 23. Assuming you average an 8% annual rate of return, how much will you have in...
-
The author conducted a study of the accuracy of weather forecasts by recording the actual high temperatures along with the high temperatures that were forecast 5 days earlier. Differences (actual...
-
Three successive resonance frequencies in an organ pipe are 1310, 1834, and 2358 Hz. (a) Is the pipe closed at one end or open at both ends? (b) What is the fundamental frequency? (c) What is the...
-
Each of the following molecules has one plane of symmetry. Find the plane of symmetry in each case: a. b. c. d. e. f. CI Me Me Br
-
Compound A has molecular formula C5H12, and monobromination of compound A produces only compound B. When compound B is treated with a strong base, a mixture is obtained containing compound C and...
-
Compare the Lewis dot structure of nitrogen and phosphorus and explain why you might expect these two atoms to exhibit similar bonding properties?
-
How to write content suitable for the cover letter that explains earned Yellow Belt credential and how the credential contributes to your strong candidacy for the position. What 4 questions would a...
-
Questions about the company Cheesecake Factory Texas Road House. General Information: 1 What is the name and ticker symbol of the corporation? 2 Where are the corporate headquarters? 3 When was the...
-
The retail industry uses inventory management techniques such as QR, EDI, barcoding and point- of-sale data capture. What does EDI stand for?

Study smarter with the SolutionInn App


