Build a truth table for each of the gate circuits in Problems 1619. P 9 DD +
Question:
Build a truth table for each of the gate circuits in Problems 16–19.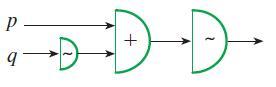
Transcribed Image Text:
P 9 DD +
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
q pV q F T...View the full answer

Answered By

RAFIKUL MALLICK
My self rafikul mallick ...i complete my B.Sc in chemistry from jhargram raj college in year 2015.....and i complete my m.sc in chemistry from vidyasagar university in year 2017...and i complete b.ed from rashbihari college of education in year 2019...now in present day i will teach many student in chemistry ...
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Build a truth table for each of the gate circuits in Problems 1619. -d 9- -D
-
Build a truth table for each of the gate circuits in Problems 1619. PE 9
-
Build a truth table for each of the gate circuits in Problems 1619. + b
-
Using the following graphs, match the market situation with the correct graph. Price 0 E1 E Q Q Quantity Graph C S 52 Price P 0 Which graph shows an increase in supply? Which graph shows a decrease...
-
In Figure two semicircular arcs have radii R2 = 7.80 cm and R1 = 3.15 cm, carry current i = 0.281 A, and share the same center of curvature C. What are the (a) Magnitude and (b) Direction (into or...
-
Journal entries, T-accounts and source documents. Production Company produces gadgets for the coveted small appliance market. The following data reflects activity for the year 2008. Production Co....
-
The stream function for an incompressible flow field is given by the equation \[ \psi=3 x^{2} y-y^{3} \] where the stream function has the units of \(\mathrm{m}^{2} / \mathrm{s}\) with \(x\) and...
-
Dave Fletcher, the general manager of North Carolina Engineering Corporation ( NCEC), thinks that his firms engineering services contracted to highway construction firms are directly related to the...
-
d Harris Company manufactures and sells a single product. A partially completed schedule of the company's total costs and c unit over the relevant range of 30,000 to 50,000 units is given below:...
-
Write a symbolic statement for each circuit shown in Problems 1015. P on off 9 on off 010
-
p q Using both switches and simplified gates, design a circuit that would find the truth values for the statements in Problems 2025 (answers are not unique).
-
Which type of industry has more rivalry, fragmented or concentrated, and why?
-
A firm that owns the stock of another corporation does not have to pay taxes on the entire amount of dividends received. In general, only 30 percent of the dividends received by one corporation from...
-
Consider the following 2015 data for Newark General Hospital (in millions of dollars): a. Calculate and interpret the profit variance. b. Calculate and interpret the revenue variance. c. Calculate...
-
What is meant by the term managed care organization (MCO)?
-
Assume that Provident Health System, a for-profit hospital, has \($1\) million in taxable income for 2016, and its tax rate is 30 percent. a. Given this information, what is the firms net income?...
-
An investment has an outlay of 100 and after-tax cash flows of 40 annually for four years. A project enhancement increases the outlay by 15 and the annual after-tax cash flows by 5. As a result, the...
-
Ralph Dewey has been charged with a violation of the Electronic Espionage Act, a federal statute that prohibits the transfer, by computer or disk or other electronic means, of a companys proprietary...
-
I frequently use NY Times and CNN and am aware of Fox News but I never use it. I visit these sites, NY Times and CNN, a few times a week whenever I have to research something or see something on...
-
Derive the ground-state term symbols for the following configurations: a. d 2 b. f 9 c. f 12
-
Two angular momenta with quantum numbers j 1 = 3/2 and j 2 = 5/2 are added. What are the possible values of J for the resultant angular momentum states?
-
The first ionization potential of ground-state He is 24.6 eV. The wavelength of light associated with the 1s2 p 1 P term is 58.44 nm. What is the ionization energy of the He atom in this excited...
-
11.13 Find m(H3O+) in a 0.10 mol/kg solution of NaCHO in water at 25C, given that Ka == 1.75 x 10-5 mol/kg for HC2H3O2 at 25C. (Hint: The acetate ion is a base and reacts with water as follows:...
-
In a three-period economy, there is a group of risk-neutral investors and a firm that has an opportunity to take on a project. At t = -1, the firm's type, either High or Low, is revealed to the...
-
Consider the financial-market imperfection model with asymmetric information. The firm needs one unit of capital to start a project that will produce y distributed uniformly over [0,2]. The firm owns...

Study smarter with the SolutionInn App


