Decide whether something is wrong with each of the graphs shown in Problems 13-17. Explain your reasoning.
Question:
Decide whether something is wrong with each of the graphs shown in Problems 13-17. Explain your reasoning.
Consider the graph shown in Figure 14.21. The potential commercial forest growth, as compared with the current commercial forest growth, is almost double (44.9 to 74.2).
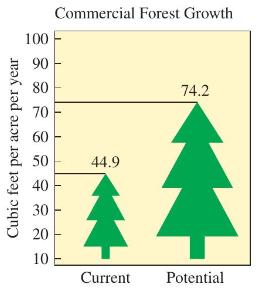
Transcribed Image Text:
Cubic feet per acre per year 100 90 80 70 60 50 40 30 20 10 Commercial Forest Growth 44.9 Current 74.2 Potential
Step by Step Answer:

This question has not been answered yet.
You can Ask your question!
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Consider an abrupt Si pn' junction that has 10" acceptors cm on the p-side and 10" donors on the n-side. The minority carrier recombination times are r=490 ns for electrons in the p-side and t=2.5 ns...
-
Decide whether something is wrong with each of the graphs shown in Problems 13-17. Explain your reasoning. The graph shown in Figure 14.23 was circulated throughout the mathematics department of a...
-
Decide whether something is wrong with each of the graphs shown in Problems 13-17. Explain your reasoning. Consider the graph shown in Figure 14.22. How much is "in the bank"? Conflicting estimates...
-
A $250 suit is on sale for $190, and a $90 pair of shoes is on sale for $65. Find the average percent decrease in price for the 2 items.
-
What is each project's initial npv without replication?
-
Do pure acids in the liquid state contain H+ ions? If not, what do such acids consist of?
-
For approximately 20 months, Robert E. McDonald perpetrated a scheme to solicit millions of dollars purportedly for a \($100\) million purchase by the RAI Entities and certain other related corporate...
-
Complete Keith's tax return including all required schedules and forms using prince edward island as province, using the fillable forms package. Taxpayers Information Taxpayer #1 Name: Keith Dox...
-
Externalities and the Environment Meyer describes the "Tragedy of the Commons." The IMF article explains how this type of problem is an example of an "externality." What is an externality? What might...
-
When a person in California renews the registration for an automobile, the bar graph shown in Figure 14.24 is included with the bill. Use this bar graph to answer the questions in Problems 18-23. The...
-
Decide whether something is wrong with each of the graphs shown in Problems 13-17. Explain your reasoning. Consider the graph shown in Figure 14.20. Chevy trucks are clearly better. 95% CHEVY FORD...
-
The accountant for Snappy photographic Supply Ltd has estimated the following activity cost pools and activity drivers for the coming year: An order for 1000 boxes of film development chemicals has...
-
Imagine you are a consultant, and you have been asked to assist Sensables's on their NPD strategic plans and Product Innovation Charter. What ideas would you suggest to them?
-
Entity C purchased two electric vehicle (EV) charging stations ($35,000 each) on January 1, 2024, for $70,000. They were originally depreciated on a straight-line basis over five years with no...
-
In the realm of file systems, how do advanced operating systems address issues related to data integrity, redundancy, and fault tolerance, incorporating concepts like journaling, RAID configurations,...
-
The company has entered into appropriate forward exchange contract to hedge .calculate the amount in kshs. that the company will receive.
-
Simplify the following radicals. 13) 375 14) 75 15) 81x5 16) 45x6y6 17) 192 8x5 18) 49x2 19) 64x18 20) 180 50x10y4 21) V-108 22) 23) 9x7y 24x12 27 24) 8y7
-
Usry Company decided to look more closely at the materials receiving activity in its factory. The driver for receiving is the number of receiving orders. The following information for a year was...
-
A circular concrete shaft liner with Youngs modulus of 3.4 million psi, Poissons ratio of 0.25, unconfined compressive strength 3,500 psi and tensile strength 350 psi is loaded to the verge of...
-
Solutions of sodium hypochlorite, NaClO, are sold as a bleach (such as Clorox). They are prepared by the reaction of chlorine with sodium hydroxide. 2NaOH(aq) + Cl 2 (g) NaCl(aq) + NaClO(aq) + H 2...
-
Copper metal reacts with nitric acid. Assume that the reaction is 3Cu(s) + 8HNO 3 (aq) 3Cu(NO 3 ) 2 (aq) + 2NO(g) + 4H 2 O(l) If 5.58 g Cu(NO 3 ) 2 is eventually obtained, how many grams of nitrogen...
-
When dinitrogen pentoxide, N 2 O 5 , a white solid, is heated, it decomposes to nitrogen dioxide and oxygen. 2N 2 O 5 (s) 4NO 2 (g) + O 2 (g) If a sample of N 2 O 5 produces 1.381 g O 2 , how many...
-
1. What are the 2 elements in developing a professional brand? Provide personal examples for each that could be beneficial in building your own professional brand. 2. Pick one of the elements of...
-
Overview: Write a response that explains your own cultural background and considers how you can improve communication for multicultural audiences Use the information and strategies from weeks 1-4 to...
-
When Olivia Lattimore and Dave Bethune from Kingston appeared on CBC's Dragons' Den, the duo believed they had a winning product. Dragon's Den is a show where entrepreneurs pitch ideas Looking for an...

Study smarter with the SolutionInn App


