Draw a scatter diagram and find (r) for the data shown in each table in Problems 25-30.
Question:
Draw a scatter diagram and find \(r\) for the data shown in each table in Problems 25-30.
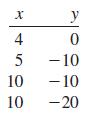
Transcribed Image Text:
X 4 5 10 10 y 0 - 10 -10 -20
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
The request is to draw a scatter diagram based on the given data and to calculate the correlation co...View the full answer

Answered By

Dinesh F
I have over 3 years of professional experience as an assignment tutor, and 1 year as a tutor trainee.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Draw a scatter diagram and find \(r\) for the data shown in each table in Problems 25-30. X 85 90 y 80 40 100 30 102 28 105 25
-
Draw a scatter diagram and find \(r\) for the data shown in each table in Problems 25-30. 10 20 30 30 50 60 y 20 48 60 58 70 75
-
Draw a scatter diagram and find \(r\) for the data shown in each table in Problems 25-30. x 0 1 2 3 4 y 25 19 16 12 10
-
Describe a procedure for solving a first-degree inequality.
-
Two waves, y1 (t) and y2 (t), have identical amplitudes and oscillate at the same frequency, but y2 (t) leads y1 (t) by a phase angle of 60o. If y1 (t) = 4cos (2 x 103t) write down the expression...
-
SSModel = 800 with SSTotal = 5820 and a sample size of n = 40 We give some information about sums of squares and sample size for a linear model. Use this information to fill in all values in an...
-
Let \(\left(N^{i}, i=1,2 ight)\) be two independent Poisson processes. Prove that \(N=N^{1}+N^{2}\) is a Poisson process. Compute the compensator of \(N\). Let \(\tau^{i}=\inf \left\{t: N_{t}^{i}=1...
-
Under what conditions is a public company allowed to dismiss its independent audit firm? Under what conditions is an independent audit firm allowed to resign as the auditor of a public company? What...
-
An activated sludge system with the MLE process (i.e., pre- anoxic system with internal recycle) has the following influent wastewater characteristics and is operated with an aerobic volume of 4000...
-
Draw a scatter diagram and find \(r\) for the data shown in each table in Problems 25-30. 1 234 1 5 8 13
-
Match the equation and correlation in Problems 19-24 with a graph. \(y=-0.7 x+2 ; r=-0.8\) 777 E. 87654321 YD 1 2 3 4 5 6 7 8 9 10 22 .. 10 1 2 3 4 5 6 7 8 9 10 8 9 10 B. 4321 8575794O ALL 21 77777 1...
-
Chadwick Industries has several manufacturing facilities that make a wide variety of industrial and consumer products. Two of the plants, River North and River South, make components for road...
-
Solve for x. Figures are not necessarily drawn to scale. Answer: x 14 42 G 42 17.5 F 12.5 E X C Submit Answer attempt 1 out of 3/problem 1 out of max 6
-
You must evaluate a proposal to buy a new milling machine. The purchase price of the milling machine, including shipping and installation costs, is $136,000, and the equipment will be fully...
-
If one developer writes code for the client program and another developer writes code for the server program, and both developers carefully follow the rules of the RFC, then the two programs will be...
-
use the java programming language with the concept of object oriented programming Family Restaurant : 1. Add Regular Menu 2. Add Special Menu 3. Show All Menu 4. Delete Regular Menu 5. Delete Special...
-
USING IntelliJ, Create project named CompSciQuiz; Implement the classes identified below; Code Specification: We are solving a classic interview problem, finding the longest prefix in an ArrayList of...
-
Describe the three types of quality performance reporting. How can managers use each report to help evaluate their quality improvement programs?
-
Calculate the change in entropy when 100 kJ of energy is transferred reversibly and isothermally as heat to a large block of copper at (i) 0 C, (ii) 50 C.
-
The typical atmospheric pressure on top of Mt. Everest (29,028 ft) is about 265 torr. Convert this pressure to (a) atm, (b) mm Hg, (c) pascals, (d) bars, (e) psi.
-
Which of the following statements best explains why nitrogen gas at STP is less dense than Xe gas at STP? (a) Because Xe is a noble gas, there is less tendency for the Xe atoms to repel one another,...
-
What change or changes in the state of a gas bring about each of the following effects? (a) The number of impacts per unit time on a given container wall increases. (b) The average energy of impact...
-
Quality management Define what is meant by internal Quality Control (IQC), external Quality Assurance (EQA) and Total Quality Management (TQM). How are IQC and EQA governed in your workplace? Provide...
-
Modern Dcor Furniture began June with merchandise inventory of 30 sofas that cost a total of $30,000. During the month, Modern Dcor purchased and sold merchandise on account as follows: June 1...
-
How might the large generation now graduating from high school affect Social Security in the future?

Study smarter with the SolutionInn App


