During the course of the following Stille coupling, the product shown was formed. How did this result
Question:
During the course of the following Stille coupling, the product shown was formed. How did this result come about?
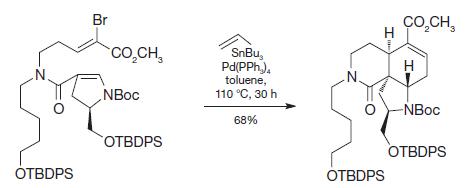
Transcribed Image Text:
OTBDPS Br CO,CH, NBoc OTBDPS SnBu Pd(PPh,), toluene, 110 °C, 30 h 68% N. OTBDPS CO,CH, H -NBoc OTBDPS H I--
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (9 reviews)
It appears that the product shown is the result of a Stille coupling reaction In a Stille coup...View the full answer

Answered By

BillClinton Muguai
I have been a tutor for the past 5 years. I have experience working with students in a variety of subject areas, including computer science, math, science, English, and history. I have also worked with students of all ages, from elementary school to college. In addition to my tutoring experience, I have a degree in education from a top university. This has given me a strong foundation in child development and learning theories, which I use to inform my tutoring practices.
I am patient and adaptable, and I work to create a positive and supportive learning environment for my students. I believe that all students have the ability to succeed, and it is my job to help them find and develop their strengths. I am confident in my ability to tutor students and help them achieve their academic goals.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Organic Chemistry
ISBN: 978-1118875766
12th Edition
Authors: T. W. Graham Solomons, Craig B. Fryhle, Scott A. Snyder
Question Posted:
Students also viewed these Sciences questions
-
During the course of 10 months, Joseph Melle sent more than 60 million unsolicited e-mail advertisements to AOL members. What charges could be brought against him?
-
During the course of serving on an audit engagement team, you discovered that your client last year recorded the receipt of cash as a sale rather than as a loan. As a result, the company's revenues...
-
During the course of auditing a telecommunication hardware company, you learned a great deal about typical credit terms and profit margins in that industry. You now have been hired by a small...
-
Assume that 3-month Treasury bills totaling $23 billion were sold in $10,000 denominations at a discount rate of 5.200%. In addition, the Treasury Department sold 6-month bills totaling $21 billion...
-
Y = 5x
-
From tables provided in this chapter, identify the NEMA designation for the following grounding-type devices used for copy machines and air conditioners requiring a 50 A, 125 V rating: a. Receptacle...
-
Use the data in Table 6.7 to compare U.S. protectionist policies with those of Japan. In what sectors are protection levels relatively equal? Where do they differ? Try to explain these patterns....
-
Tammy Krause is the new owner of Tammys Computer Services. At the end of July 2012, her first month of ownership, Tammy is trying to prepare monthly financial statements. She has the following...
-
A 22 kg dolphin moving in the positive direction decelerates from 12.5 to 8.5 m/s in 2.5 s to join another dolphin in play. What average force, in newtons, was exerted to slow him if he was moving...
-
A cantilever rod is loaded as shown in the figure below. If the tensile yield strength of the material is 300 MPa determine the rod diameter using (a) Maximum principal stress theory (b) Maximum...
-
Olestra is a fat substitute patented by Procter and Gamble that mimics the taste and texture of triacylglycerols. It is calorie-free because it is neither hydrolyzed by digestive enzymes nor absorbed...
-
Propose mechanisms for the following reactions. (a) (b) Br OH cat. HSO4 Br
-
The following is a cumulative percent distribution for data on minutes spent exercising in a population-based study with college students. Based on the data in this table, in which interval does the...
-
A citys General Fund records expenditures for uncollectable general government property tax revenues? a. When the property taxes are levied b. When property taxes are written off as uncollectable c....
-
A 130 g ball is dropped from a height of 12 m. Determine the following: a. The velocity of the ball when it hits the ground. b. The velocity of the ball at a height 1 m above the ground.
-
How do asymmetric information and behavioral economics influence the strategic decision-making process, ultimately shaping firms' ability to outmaneuver competitors and secure superior market...
-
An unlevered firm has a cost of capital of 16% and earnings before interest and taxes of $225,000. A levered firm with the same operations and assets has both a book value and a face value of debt of...
-
Assume firms within an industry do not use price to compete with. In general, they do not tend to challenge rivals by dropping price. The competition is not trying to take market share through lower...
-
If U and Ware subspaces of V, define their intersection U W as follows: U W = {v|v is in both U and W} (a) Show that U W is a subspace contained in L and W. (b) Show that U W {0} if and only if...
-
According to a recent survey, 40% of millennials (those born in the 1980s or 1990s) view themselves more as spenders than savers. The survey also reveals that 75% of millennials view social...
-
Show the products of thesereactions: Bra Br2 a) CH,CH,CH=CH2 b) CH Cl2 CH Cl2 excess Cl2 CH-Cl2 Brz CCI4 d)
-
Show all of the steps, including stereochemistry, in the mechanism for thisreaction: Br + Br2 Br
-
Explain which of these compounds has the faster rate of reaction withBr2: Ph Ph - c=CH, or .
-
Houston stock is selling for $47 and has the following six-month options outstanding. Strike Price Option Market Price Call Option $45 $5 Call option $50 $2 a. Which option(s) is (are) in the...
-
A regional electrical distributor currently has 1,000 customers who buy an average of $5,000 per year, generating a 50% margin. From experience, the company knows that 20% of its customers will not...
-
Suppose you observed that one-year T-bills are trading with a yield to maturity (YTM) of 4.75%. The yield spread between AAA and BB rated corporate bonds is 130 basis points. The maturity yield...

Study smarter with the SolutionInn App


