For each of the following complexes, determine the oxidation state of the metal and the total number
Question:
For each of the following complexes, determine the oxidation state of the metal and the total number of valence electrons it possesses.
(a)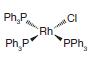
(b)
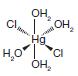
(c)
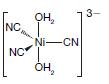
Transcribed Image Text:
Ph.P H Ph P CI PPh mee Rh
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 60% (5 reviews)
Answer a The oxidation state of the metal in the complex is 3 a...View the full answer

Answered By

Justin Akengo
I am writing in application for the tutor position with your organisation. I am experienced in tutoring students of all abilities and I believe I am the ideal candidate for this position.
I work with students of all ages, from elementary school to college level. Whether the subject is science, Mathematics or basic study skills, I break material down into easy-to-understand concepts. In your job posting, you asked for someone who can tutor in a variety of subjects. I am comfortable explaining calculus to a college student or working with a kindergartener on spelling fundamentals.
Below are just a few core skills and qualifications I posses as a tutor;
Adept at creating study materials in a variety of academic subjects to help students improve their test scores and GPAs.
Strong interpersonal skills in working with students to help them achieve and succeed.
Have written study books adopted by a high school and a college to help students improve their skills in English and mathematics.
Have won several “Tutor of the Year” awards for work with high school and college students.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Organic Chemistry
ISBN: 978-1118875766
12th Edition
Authors: T. W. Graham Solomons, Craig B. Fryhle, Scott A. Snyder
Question Posted:
Students also viewed these Sciences questions
-
For each of the following complexes, determine the oxidation state of the transition-metal atom. a. [CoCl(en)2(NO2)]NO2 b. [PtCl4]2 c. K3[Cr(CN)6] d. [Fe(H2O)5(OH)]2+
-
State the research problems for each of the following management decision problems. a. Should a new product be introduced? b. Should an advertising campaign that has run for three years be changed?...
-
State the appropriate nonparametric test for each of the following examples. (a) A physiological psychologist compares differences in the amount of chemical activity in a certain brain region between...
-
1. Prepare a schedule of cost of goods manufactured for Denim Bones for the year ended December 31, 2024. 2. Prepare an income statement for Denim Bones for the year ended December 31, 2024. 3. How...
-
The percentages of Americans who are satisfied with the way things are in the United States are shown in Table 59 for various years. Let p be the percentage of Americans at t years since 1990 who are...
-
Thermal convection is linked mostly to (a) radiant energy. (b) fluids. (c) insulators. (d) all of these
-
During the year ended 30 June 2025, Trendline Pty Ltd incurred the following costs. The company charges factory overhead costs to work in process inventory and finished goods inventory, using an...
-
Suppose a perfectly competitive industry can produce Roman candles at a constant marginal cost of $10 per unit. Once the industry is monopolized, marginal costs rise to $12 per unit because $2 per...
-
Describe the overall process for effectively using technology within an organization. Consider such aspects as strategic alignment, planning, centralized systems, data governance, cybersecurity, the...
-
1. Imagine that an agency or business has asked for help in gathering data about the number of sports anglers who fish off the coast of Georgia. What advice would you give about sampling? What method...
-
What is the product of the following SuzukiMiyaura coupling? Br B(OR)2 + Pd catalyst Base
-
In The Chemistry of Complex Cross Couplings we showed a Heck reaction used in the synthesis of ecteinascidin 743, a natural compound with anticancer activity isolated from the marine tunicate...
-
Customer-based inventory management strategies allow the use of different availability levels for specific customers. Discuss the rationale for such a strategy. Are such strategies discriminatory?...
-
Distinguish the drift speeds of conduction electrons from their random-motion speeds, including relative magnitudes. If the current carried by a conductor is tripled, what happens to (a) the charge...
-
A tariff levied on a good produced in a small nation with an inelastic supply that maximizes the gain to a large nation is called a(n)
-
Think of the etiology, symptoms, diagnosis and treatment of children with Down Syndrome and Autism Spectrum Disorder. Explain the importance of early intervention, specifically as it relates to...
-
The ideal (theoretical) pressure increase across a centrifugal pump is 10m. The radial velocity through the pump is constant at 2m/s. The exit angle of the impeller vanes is 60 and the water enters...
-
While competitors like Compaq did not. HP acquired Compaq in 2002. How would the just-in-time manufacturing model affect the Inventory Turnover ratio?
-
Let V denote the set of ordered triples (x, y, z) and define addition in V in R3. For each of the following definitions of scalar multiplication, decide whether V is a vector space. (a) a(x, y, z) =...
-
Write a declaration for each of the following: a. A line that extends from point (60, 100) to point (30, 90) b. A rectangle that is 20 pixels wide, 100 pixels high, and has its upper-left corner at...
-
Predict the positions of the major absorption bands in the IR spectra of thesecompounds: CH30 CH-NH, a) CH3CH=CHCCH; c) b) C-H NO, e) CH-CH,C3- d) CH,CH,CH,OH CH3
-
Explain how IRD spectroscopy could be used to distinguish between these compounds: CH;CH,C=CH a) CH,CH,CH=CH; and CH3 H- b) CH, nd CH3 CCH3 CH c) and d) CH.CH,CH,C,NH, and CH.CH,NHCH,CH,
-
List the positions of the important absorption bands in the IR spectra of these compounds: b) H,H-CH-C3DN ) -H,H,NH> CCH,CH3 c) CH;CH,COCH;CH,CH3 d)
-
Water flows through a pipe section that tapers from an internal diameter of 200 mm down to 100 mm over a distance of 1 m. Determine the pressure drop over the section for a flow of 0.06 m3s-1. The...
-
Calculate the binding energy per nucleon for Be, 59 Co, 208 pb, and 195 pt. (For the atomic masses, see this table. Enter your answers to at least two decimal places.) (a) Be MeV/nucleon 59 Co...
-
A.Final ProjectTeamProjectCustom Schema: 200 points Extra Credit Group of 3 people : Designer and Developer ( ERD, tables, data ) Team Leader/Power User ( Business Questions/Queries, Real Life...

Study smarter with the SolutionInn App


