Write a mechanism for the following reaction. cat. HSO4 CHOH OCH 3
Question:
Write a mechanism for the following reaction.
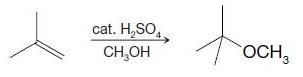
Transcribed Image Text:
cat. H₂SO4 CH₂OH OCH 3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (12 reviews)
The mechanism for the following reaction is as follows Step 1 Pro ton ation of the al...View the full answer

Answered By

SHINKI JALHOTRA
I have worked with other sites like Course Hero as a tutor and I have great knowledge on IT skills.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Organic Chemistry
ISBN: 978-1118875766
12th Edition
Authors: T. W. Graham Solomons, Craig B. Fryhle, Scott A. Snyder
Question Posted:
Students also viewed these Sciences questions
-
Write a mechanism for the following reaction and indicate the type of reaction that takes place for each of them. Also give the number of p electrons and include designations of conrotatory or...
-
Write a radical chain mechanism for the following reaction (a reaction called the Hunsdiecker reaction). Br Br CO2
-
Write a mechanism for the reaction shown in eq. 10.35. room C-OCH, +HCl (10.35) temp benzoyl chloride methyl benzoate
-
Calculate the following ratios from the data given below: 1. Debt ratio 2. Debt service coverage multiples 3. Interest coverage Balance Sheet 12/31/2014 Assets Cash..$575,000 Short-term...
-
Explain how local content requirements, administrative delays, and currency controls restrict trade.
-
Explain how the partnership accounts for the sale by a partner of a portion of his partnership interest to another individual.
-
In a non-self-starting method, we need to generate the value of the following quantity using the finite difference approximations of \(\dot{x}_{i}\) and \(\ddot{x}_{i}\) : a. \(\dot{x}_{-1}\) b....
-
Your supervisor, Jesse Baker, has asked you to begin working on data design tasks for the new information system, which will be implemented as a relational database. You will need to identify the...
-
P = 780 kN P = 780 kN 3 m X 3m 3 m X 3m 0.2 m 0.2 m Sand 1.4 m 2.0 m 1.4 m Clay 1.2 m Bedrock
-
SHL Americas provides a unique, global perspective of how talent is measured in its Global Assesment Trends Report. The report presents the results of an online survey conducted in late 2012 with HR...
-
Write a mechanism that shows how 2,3-dimethyl-2-butanol is formed in the acid-catalyzed hydration of 3,3-dimethyl-1-butene.
-
cis-4-Bromocyclohexanol t - Buo / t - Buohracemic C 6 H 10 O (compound C) Compound C has infrared absorption in the 16201680 cm 1 and in the 35903650 cm 1 regions. Draw and label the (R) and (S)...
-
In Figure, does unemployment change more rapidly when it is increasing, or when it is decreasing? Based on previous experience, when will the unemployment rate again reach 4% after the 20082009...
-
These items are taken from the financial statements of Pronghorn Corp. at December 3 1 , 2 0 2 5 . Buildings $ 1 2 1 , 7 0 0 Accounts receivable 1 4 , 5 0 0 Prepaid insurance 5 , 3 8 0 Cash 1 3 , 6 2...
-
Constants The aurora is caused when electrons and protons, moving in the earth's magnetic field of 5.0 x 105T, collide with molecules of the atmosphere and cause them to glow. Part B What is the...
-
5. The picture to the left shows a bucket connected to a massless rope, which runs over a massless, frictionless pulley. A man standing inside the bucket pulls the rope downwards in order to raise...
-
what cognitive alchemy does the visionary transmute the leaden constraints of the present into the gilded opportunities of tomorrow, orchestrating symphonies of innovation amidst the cacophony of...
-
Futura Company purchases the 4 0 , 0 0 0 starters that it installs in its standard line of farm tractors from a supplier for the price of $ 8 . 4 0 per unit. Due to a reduction in output, the company...
-
The following accounts receivable information is for Rouge Company: Required: With the big increase in Allowance for Bad Debts in 2009, Rouge Company is concerned that the creditworthiness of its...
-
In the operation of an automated production line with storage buffers, what does it mean if a buffer is nearly always empty or nearly always full?
-
Show the most stable conjugate base of these compounds: a) CHCHOH i b) HOCCHCHOH c) HNCHCHOH
-
Which of these species can behave as a Lewis acid? CI a) Cl-B CI H b) H-C-H H + c) CHCH
-
Which of these species can behave as a Lewis base? a) CH0CH,CH, CH3 d) CH3NH + b) CH3CHCH3 0: I.. e) CHCOH c) CH3NH
-
A start-up has been approached by a Venture Capital investor, who would like to invest $700,000 into the business. The start-up's CEO is preparing for the term sheet negotiations, and wants to know...
-
The following selected accounts and their current balances appear in the ledger of Aqua Co. for the fiscal year ended June 30, 20Y8: Cash $ 83,500 Sales $3,625,000 Accounts Receivable Merchandise...
-
You will make a post giving your point-of-view (PoV) on a brand's activities and/or marketing campaign. Give your PoV on what brands can and should do to ensure they are creating a great digital...

Study smarter with the SolutionInn App


