Which of these species can behave as a Lewis base? a) CH0CH,CH, CH3 d) CH3NH + b)
Question:
Which of these species can behave as a Lewis base?
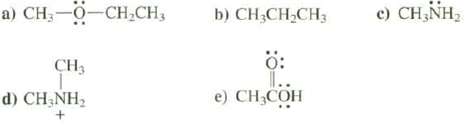
Transcribed Image Text:
a) CHẠ—0—CH,CH, CH3 d) CH3NH₂ + b) CH3CH₂CH3 0: I.. e) CH₂COH c) CH3NH₂
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 55% (20 reviews)
According to the Lewis definition a ...View the full answer

Answered By

MICHAEL KICHE
I was employed studypool for the first time in tutoring. I did well since most of my students and clients got the necessary information and knowledge requested for. I always submitted the answers in time and followed the correct formatting in answering eg MLA or APA format,
Again I worked with the writers bay where I did writing and got many clients whom we worked with so closely. They enjoyed every single service I delivered to them. My answers are always correct.
4.70+
13+ Reviews
53+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Which of these species can behave as a Lewis acid? CI a) Cl-B CI H b) H-C-H H + c) CHCH
-
Indicate whether each of these species can act as an acid, a base, or both: H +1 a) H-N-H 1 H H e) H-C-O-H H b) H-O-H H: H TI f) H-C-C-C-H H H c) H-C-H H H d):Ci : g) H-O-C-:
-
Which species is more stable? a. b. c. d. e. f. CH10. CHOH,or or CH3CCHCH2CH or CH CCHCCH CH3CHCH2CCH3 or CH3CH CHCCH3 NH CHICHCHsor CHCNH CH CHCH3 or CHCNH2 CH2 CH C-CH or CH C-CH CH3 CH3 or
-
Identifying the leadership strategies that were employed by the organization described in the case study, how they were employed, and any additional strategies that could be employed to reflect...
-
Zappy Kitchens manufactures a range of household products. Susan Masters, the company's management accountant, plans to implement a standard costing system. Masters has collected information from...
-
Use the following data to answer the following questions (all figures are in billions of dollars): Item Amount Government purchases of goods and services.$1,721.6 Exports... 1,096.3 Receipts of...
-
A Piper Cub airplane has a gross weight of \(1750 \mathrm{lb}\), a cruising speed of \(115 \mathrm{mph}\), and a wing area of \(179 \mathrm{ft}^{2}\). Determine the lift coefficient of this airplane...
-
The Mark Goodwin Resort is an elegant summer resort located in a remote mountain setting. Guests visiting the resort can fish, hike, go horseback riding, swim in one of three hotel pools, or simply...
-
You think the political solutions presented by the author are still valid or are other strategies required? "A System at Risk is a comprehensive analysis of the transportation dilemma in the United...
-
A national bank has developed a predictive model for identifying customers who are more likely to accept a credit card offer, if a customer is predicted to accept the credit card offer, he or she is...
-
Show the most stable conjugate base of these compounds: a) CHCHOH i b) HOCCHCHOH c) HNCHCHOH
-
Calculate the pKa for these compounds. a) HCOH (K 1.75 X 104) b) CHCH3 (K = 10-50)
-
A stream of water flowing at a rate of 1.2 m3/min and moving with a velocity of magnitude v at both A and B is deflected by a vane welded to a hinged plate. The combined mass of the vane and plate is...
-
Should a board member have personal liability insurance coverage?
-
Discuss in what ways the rights and responsibilities of patients and employees are alike.
-
Under what circumstances should a healthcare professional self-insure?
-
What are the distinct parts of an insurance policy?
-
Describe the purpose and process of utilization review.
-
This problem asks you to do your own growth accounting exercise. Using data since 1960, make a table of annual growth rates of real GDP, the capital stock, and civilian employment. Assuming...
-
(8%) Problem 6: A student attaches a f= 3.5 kHz oscillator to one end of a metal rail of length L = 25 m. The student turns on the oscillator and uses a piezoelectric gauge at the other end to...
-
Sketch the region in the xy-plane. {(x, y) ||x| 2}
-
Give structures that fit the following descriptions: (a) An , -un-saturated ketone, C6H8O (b) An -dike tone (c) An aromatic ketone, C9H10O (d) A diene aldehyde, C7H8O
-
Predict the products of the reaction of (i) phenyl acetaldehyde and (ii) acetophenone with the following reagents: (a) NaBH4 then H3O+ (b) Tollens reagent (c) NH2OH, HC1 catalyst (d) CH3MgBr, then...
-
How would you prepare the following substances from 2-cyclohexcnone? More than one step may be required. .CH (a) (b) (d) (c) CSH5 (Two ways)
-
Frank is 82 years old.His wife died 5 years ago.He died and left the following assets: Principal residence 859,000 (individually owned: ACB 350,000) RRIF $235,000 (Beneficiary adult daughter ACB...
-
Assume that Pope Enterprises held a $10,000, 10 percent, six-month note signed by Mary Drew. On December, 1, 2015, the maturity date, Drew dishonored the note. At this point, Drew owes a total of...
-
5. Consider the shape shown on the following graph and the brute force method of solving the convex hull problem 2 -2 -1 0 a. Label all the points that will be provided to the algorithm as input. You...

Study smarter with the SolutionInn App


