a. Which of the following reactions will not give the carbonyl product shown? 1. 2. 3. 4.
Question:
a. Which of the following reactions will not give the carbonyl product shown?
1.
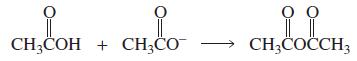
2.
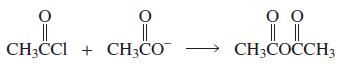
3.
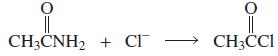
4.
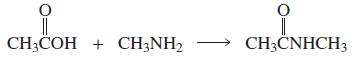
5.
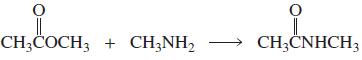
6.
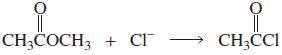
7.
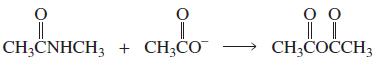
8.
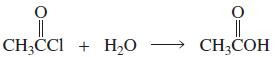
9.
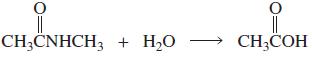
10.
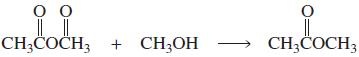
Transcribed Image Text:
LI CH;COČCH3 CH;COH + CH;CO
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 60% (10 reviews)
1 this reaction is not happening because acetate ion always abstracts the proton then becomes stable This is a reversible reaction CH 3 COCOOH CH 3 CO...View the full answer

Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Which of the following reactions occurs most rapidly? Why? a. b. c. Br - H20 C(CH3)3 C(CH3)a (CH)a C(CH3)3 Br - H20 CH33 C(CH3)3
-
Which of the following reactions and decays are possible? For those forbidden, explain what laws are violated. (a) + p n + 0 (b) + + p n + 0 (c) + + p p + e0 (d) p e+ + vp (e) + e+ + vp (f) p n + e...
-
Which of the following reactions are possible, and by what interaction could they occur? For those forbiddevn explain why. (a) + p K0 + p + 0 (b) K + p 0 + 0 (c) K+ + n + + 0 + y (d) K + 0 + 0 + +...
-
A water sprinkler sprays water over a distance of 30 feet while rotating through an angle of 135. What area of lawn receives water? 135 30 ft
-
The graph shows the effect of pH on quenching of luminescence of tris(2,2 bipyridine)Ru(II) by 2,6-dimethylphenol. The ordinate, K SV , is the collection of constants, k q /(k e + k d ), in the...
-
Consider preparing a DSS for which certainty factors are relevant. This may be a class project, an example with which you are familiar, or a hypothetical example. What issues need to be tracked with...
-
After receiving complaints from readers, your campus newspaper decides to redesign its front page. A new format B is developed and tested against the current format, A. A total of 100 students are...
-
Gifford, Lawrence, and Ma share equally in net income and net losses. After the partnership sells all assets for cash, divides the losses on realization, and pays the liabilities, the balances in the...
-
Explain how tax-benefit linkages, spillovers, and economies of scale influence fiscal federalism (that is, what each of those implies in terms of the optimal division of responsibilities across the...
-
You are a senior auditor with Rodriguez & Jones, a small auditing firm located in Canterbury, an eastern suburb of Melbourne, Victoria. Your team has been assigned to the audit of a new client,...
-
An oil obtained from coconuts is unusual in that all three fatty acid components are identical. The molecular formula of the oil is C 45 H 86 O 6 . What is the molecular formula of the carboxylate...
-
a. Which of the following reactions will not give the carbonyl product shown? 1. 2. 3. 4. 5. 6. 7. 8. 9. 10. b. Which of the reactions that do not occur can be made to occur if an acid catalyst is...
-
In an audit sampling application, an auditor a. Performs procedures on all items in a balance and makes a conclusion about the entire balance. b. Performs procedures on less than 100 percent of the...
-
What is management audit?
-
What is periodical audit? What are the advantages and disadvantages of periodical audit? Distinguish between continuous audit and periodical audit.
-
(a) Distinguish between internal audit and statutory audit. (b) Can the statutory auditor rely upon the internal audit in carrying out his function as a statutory auditor?
-
Explain the following statement (a) Vouching is the essence of auditing. (b) In vouching payments, the auditor does not merely seek proof that money has been paid away.
-
What precautions are to be taken in the application of internal check system?
-
In 1974, interest rates were 7.782% and the rate of inflation was 12.299% in Canada. What was the real interest rate in 1974? How would the purchasing power of your savings have changed over the year?
-
Frontland Advertising creates, plans, and handles advertising campaigns in a three-state area. Recently, Frontland had to replace an inexperienced office worker in charge of bookkeeping because of...
-
Use electronegativities to predict the direction of the dipole moments of the following bonds. (a) C-CI (b) C-O (c) C-N (d) C-S (e) C-B (f) NCl (g) NO (h) NS (i) NB (j) B-CI
-
The following compounds can all react as acids. (a) For each compound, show its conjugate base. Show any resonance forms if applicable. (b) Rank the conjugate bases in the order you would predict,...
-
The following compounds can all react as bases. (a) For each compound, show its conjugate acid. Show any resonance forms if applicable. (b) Rank the conjugate acids in the order you would predict,...
-
2. Consider the metal ion complex ferricyanide, [Fe(CN)6], obtained when potassium hexacyanoferrate(III) is dissolved in solution. a) Predict the number and intensity of all its ligand field (d-d)...
-
16) Chris, Bob, and Matt decide to help each other study during the final exams. Chris's favorite subject is chemistry, Bob loves biology, and Matt knows his math. Each studies his own subject as...
-
Match the column A with column B and Give Supported explanations. E Charge 1 Charge 2 Distance Apart Force Attracted/Repelled / Neither Trial 1 -4 C 4 cm Trial 2 -4 C -2 C 4 cm Trial 3 -4C -4C 4 cm...

Study smarter with the SolutionInn App


