Are the following pairs of structures resonance contributors or different compounds? a. b. CH 3 CH=CHCHCH=CH 2
Question:
Are the following pairs of structures resonance contributors or different compounds?
a.
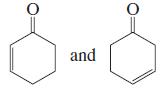
b. CH3CH=CHĊHCH=CH2 and CH3ĊHCH=CHCH=CH2
c.

d.
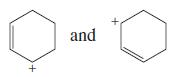
e. CH3C+HCH=CHCH3 and CH3CH=CHCH2C+H2
Transcribed Image Text:
and
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 71% (14 reviews)
8888 Different compounds a b HC CH CH CH CH CH2 and HC CH CH CH CH CH and d ...View the full answer

Answered By

Shyam Kumar
I have done B.Tech in chemical engineering .I have teaching high school level students and for engineering/medical entrance exams like IIT,NEET etc I have very good experience of dealing with all kinds of students weaker and brilliant and how to develop interest among students by representating the topics in unique way so that they can learn without stress and enjoy the subject.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Give the relationship between the following pairs of structures. The possible relationships are: same compound cis-trans isomers constitutional isomers (structural isomers) not isomers (different...
-
Give the relationships between the following pairs of structures. The possible relationships are: same compound, cis-trans isomers, constitutional (structural) isomers, not isomers (different...
-
Determine whether the following pairs of structures are actually different compounds or simply resonance forms of the same compounds. (a) (b) (c) (d) (e) (f) (g) (i) (j) and O- and and O- CH C H and...
-
How to respond to this response The effective utilization of data is integral to enhancing efficiency and making informed decisions?
-
Calculate the Doppler linewidth for the 589-nm line of Na and for the 254-nm line of Hg, both at 2 000 K.
-
Determine the distance y to the center of gravity of the homogeneous rod. 1 m 2 m Fy 2r
-
Develop the questionnaire in Internet Exercise 8-21 using an electronic questionnaire design package that is available for free (see the websites mentioned in this chapter in the subsection entitled...
-
1. Why would a $500,000 salary cap prompt personnel to leave for other banks? 2. Was the stripping of Lewiss chairmanship a significant move on the part of BoA shareholders? 3. How could John Thain...
-
After taking a look at Fisher-Price's website, choose two effective and two ineffective KPIs for this company that would be aligned with the sales and marketing goals. For all the KPIs, you are to...
-
Suppose college mens heights are approximately normally distributed with a mean of 70.0 inches and a population standard deviation of 3 inches. What height is at the 20th percentile? Include an...
-
Draw resonance contributors for the following ions: a. b. c. d.
-
The triphenylmethyl cation is so stable that a salt such as triphenylmethyl chloride can be isolated and stored. Why is this carbocation so stable? C. triphenylmethyl chloride
-
On December 31, 1776, Rhode Island established wage controls to limit wages to 70 a day for carpenters and 42 a day for tailors. a. Are these wage controls a price ceiling or a price floor? Why might...
-
Construct a multi-attribute model to compare three different restaurants for an important celebration in your family. Apply the two different decision rules and determine the choices that arise from...
-
Explain the meaning of customer loyalty and customer engagement.
-
This case studies a successful marketplace to prompt learning about what makes such a marketplace effective. It also illustrates the importance of online bidding in some industries. The Covisint...
-
How can customer choice between services in their consideration set be modeled?
-
Explain why services tend to be harder for customers to evaluate than goods.
-
What kind of real option does the XC-900 machine (see Problem 19) provide to Buckingham?
-
(a) What do data breach notification laws require? (b) Why has this caused companies to think more about security?
-
Give the m/z values of the fragment ions expected from b - type fragmentation of an M + 1 ion of the peptide N - F - E - S - G - K
-
Give the curved-arrow mechanism for the formation of each of the following fragment ions in Fig. 26.5 from an M + I ion. The fragment at m/z = 551.94
-
Using the curved-arrow notation, write in detail the mechanisms for the reactions in (a)Eq. 26.41a
-
Complete the Price, Advertising, and Profitability chart. a . ?Notice that the series for $ 2 5 , 0 0 0 , 0 0 0 ?is already on the chart. b . ?Add the series for advertising budgets $ 5 0 , 0 0 0 , 0...
-
Solve for the missing information designated by "?" in the following table. (Use 365 days in a year. Round the inventory turnover ratio to one decimal place before computing days to sell. Round days...
-
Grove Media plans to acquire production equipment for $800,000 that will be depreciated for tax purposes as follows: year 1, $320,000; year 2, $180,000; and in each of years 3 through 5, $100,000 per...

Study smarter with the SolutionInn App


