Question: Explain why the ether obtained by treating an optically active alcohol with PBr 3 followed by sodium methoxide has the same configuration as the alcohol,
Explain why the ether obtained by treating an optically active alcohol with PBr3 followed by sodium methoxide has the same configuration as the alcohol, whereas the ether obtained by treating the alcohol with tosyl chloride followed by sodium methoxide has a configuration opposite that of the alcohol.
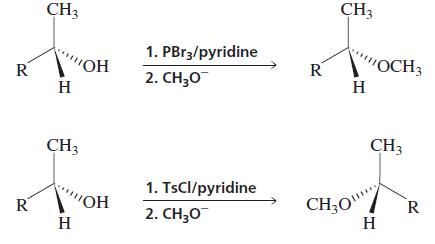
CH3 CH3 1. PBr3/pyridine R OH R "OCH3 H 2. H,0 H CH3 CH3 1. TsCl/pyridine CH;O" H R VOH R 2. CH;0 H
Step by Step Solution
3.26 Rating (161 Votes )
There are 3 Steps involved in it
When the ether obtained by treating the optically active alcohol with PBr 3 followed by s... View full answer

Get step-by-step solutions from verified subject matter experts


