Fill in any unshared electrons that are missing from the following linebond structures: (a) H3C CH3 Dimethyl
Question:
Fill in any unshared electrons that are missing from the following linebond structures: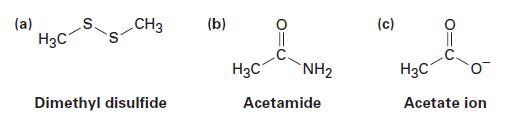
Transcribed Image Text:
(a) H3C CH3 Dimethyl disulfide (b) H3C NH₂ Acetamide (c) H3C Acetate ion
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (4 reviews)
A The given compound is Dimethylsulphide As you can see each s...View the full answer

Answered By

Samee Ullah
Algebra, Linear algebra, calculus, accounting, marketing, statistics, programming, real estate, writing, human resource management, business communication, Engineering: civil, chemical, electrical, mechanical, aerospace, building
Linguistics: sociolinguistics, applied linguistics, music, social sciences, biology, chemistry: all types, Thermodynamics, mechanics, modern physics, quantum physics, metaphysics, biology.
Feel free to contact us for all these subjects,; for quality, and best responses. Thankyou
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Fill in any unshared electron pairs that are missing from the following formulas: (a) (CH3CH2)2NH (b) (c) CH3CH2SCH2CH3 (d) CH3OCH2CH2OH CH, O=U
-
Fill in any nonbonding valence electrons that are missing from the followingstructures: CH (b) (c) (a) " NH2 - Dimethyl disulfide Acetate ion Acetamide
-
Add any missing unshared electron pairs (if any), then, using curved arrows to show the shifts in electrons, write the contributing resonance structures and resonance hybrid for each of the...
-
Under a plan of complete liquidation, Coast Corporation distributes land with a $300,000 adjusted basis and a $400,000 FMV to William, a 25% shareholder. William has a $200,000 basis in his Coast...
-
Determine whether each of the following is true or false. Assume that each organization is a private, not-for-profit entity. It is possible that an item need not be reported on the hospitals...
-
J. C. Penny produced a T-shirt emblazoned with a ferocious red and blue eagle beneath the words American Made. However, a small label inside revealed that the shirt was made in Mexico. In response to...
-
Bert C. Roberts Jr. was chairman of WorldComs board of directors. Immediately before that, he had been chairman of MCI, which WorldCom acquired on September 14, 1998, in a transaction valued at...
-
Fogelberg Corporation is a regional company which is an SEC registrant. The corporations securities are thinly traded on NASDAQ (National Association of Securities Dealers Quotes). Fogelberg has...
-
A. Find the value of x. B. 1. 2. 61 = x ( mod 7) 71 = x ( mod 8) 3. 78 x 3 (mod 5) 4. 89 = (x+3)( mod 4) 1. What is the remainder when 2021 2021 2020? is divided by
-
Use the electronegativity values in Figure 1.14 to predict which of the indicated bonds in each of the following sets is more polar. Tell the direction of the polarity in each. (a) ClOCH 3 or ClOCl...
-
Amide ion, H 2 N - , is a stronger base than hydroxide ion, HO - . Which is the stronger acid, H 2 NOH (ammonia) or HO-H (water)? Explain.
-
1. During the year ended 30 June 2019, French Horn Ltd made cash sales of $100 000, credit sales of $200 000 ($50 000 of which were still to be collected at year-end), and received $25 000 owing from...
-
Your company has been a one of the few producers of your product. You multisource one of your key inputs from nearly a dozen small suppliers, and have been nearly the only market for some of the...
-
Find the net charge of a system consisting of 6.21106 electrons and 7.8710 protons. Express your answer in coulombs. View Available Hint(s) Qnet = Submit Part B t O ? C Find the net charge of a...
-
Assume market demand characterized by MPB(Q)=168-2Q (MPB=marginal private benefit, and is the same as the price in the demand function), and market supply characterized by MPC(Q)=12+2Q (MPC is the...
-
Bryan paid $96.00 for an item at the store that was 20% off the original price. What was the original price? The original price for the item was $ Round your answer to the nearest cent.
-
Express the total-spin states |1, 1) and |1, 1) (with respect to the z-axis, as usual) of a system made of two spin- particles in terms of the product states | +x, +x), |+x, x), |x, +x), and | x,...
-
The World Series of baseball is to be played by team A and team B. The first team to win four games wins the series. Suppose that team A is the better team, in the sense that the probability is 0.6...
-
What is a content filter? Where is it placed in the network to gain the best result for the organization?
-
The principle of least motion, which states that the reaction that involves the least change in atomic positions or electronic configuration (all else being equal) is favored, has been suggested to...
-
Investigation has shown that cyclobutadiene is actually a rectangular molecule rather than a square molecule. In addition, it has been established that there are two different...
-
Explain why HBr should be used to generate the benzenediazonium salt if bromobenzene is the desired product of the Sandmeyer reaction and HCl should be used if chlorobenzene is the desired product.
-
All of the capacitors in the figure below are 6.6 F. What is the equivalent capacitance of this network? 9V
-
10. Two charges are evenly spaced in 2 cm intervals on the x-axis: q = 10 nC and q = -3 nC. How much work is required to bring a positive charge of q3 = 4 nC from a point at infinity to point A if...
-
An air-filled capacitor consists of two parallel plates, each with an area of 7.60 cm, separated by a distance of 2.00 mm. If a 16.0 V potential difference is applied to these plates, calculate the...

Study smarter with the SolutionInn App


