Use the electronegativity values in Figure 1.14 to predict which of the indicated bonds in each of
Question:
Use the electronegativity values in Figure 1.14 to predict which of the indicated bonds in each of the following sets is more polar. Tell the direction of the polarity in each.
(a) ClOCH3 or ClOCl
(b) HOCH3 or HOCl
(c) HOOCH3 or (CH3)3SiOCH3
Fig 1.14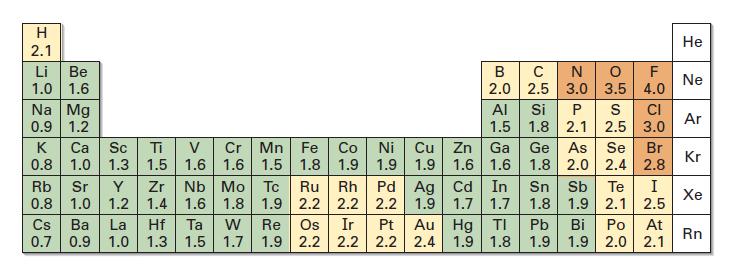
Transcribed Image Text:
H 2.1 Li Be 1.0 1.6 Na Mg 0.9 1.2 Cr Mn Fe Co Ni Cu 1.6 1.6 1.5 1.8 1.9 1.9 1.9 K Ca Sc Ti V 0.8 1.0 1.3 1.5 Rb Sr Y Zr 0.8 1.0 1.2 1.4 Cs Ba La Hf 0.7 0.9 1.0 1.3 B C N O F 2.0 2.5 3.0 3.5 4.0 Ta W 1.5 1.7 AI 1.5 Zn Ga 1.6 1.6 1.9 1.7 1.7 Nb Mo Tc Ru Rh Pd Ag Cd In 1.6 1.8 1.9 2.2 2.2 2.2 Re Os Ir Pt 1.9 2.2 2.2 2.2 Au Hg ΤΙ 2.4 1.9 1.8 Si P S CI 1.8 2.1 2.5 3.0 Ge As 1.8 2.0 2.4 Se Br 2.8 Sn Sb Te 1.8 1.9 2.1 Pb 1.9 I 2.5 Bi Po At 1.9 2.0 2.1 He Ne Ar Kr Xe Rn
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (5 reviews)
To predict the polarity of each bond we need to compare the electronegativity values of the atoms involved in the bond The more significant the differ...View the full answer

Answered By

Surendar Kumaradevan
I have worked with both teachers and students to offer specialized help with everything from grammar and vocabulary to challenging problem-solving in a range of academic disciplines. For each student's specific needs, I can offer explanations, examples, and practice tasks that will help them better understand complex ideas and develop their skills.
I employ a range of techniques and resources in my engaged, interesting tutoring sessions to keep students motivated and on task. I have the tools necessary to offer students the support and direction they require in order to achieve, whether they need assistance with their homework, test preparation, or simply want to hone their skills in a particular subject area.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Use the electro negatively table (Figure) to predict which bond in each of the following sets is more polar, and indicate the direction of bond polarity for each compound. (a) H3C ? C1 OR C1 ? C1 (b)...
-
In Example 11 on ESP, John Doe had to predict which of five numbers was chosen in each of three trials. Doe did not actually have ESP. Explain why this experiment satisfies the three conditions for...
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-5. Ivan's grandfather died and left a portfolio of municipal bonds. In 2012, they pay Ivan...
-
Show that for an integer n > 2, the period of the decimal expression for the rational number is at most n - 1. Find the first few values of n for which the period of - is equal ton- 1. Do you notice...
-
The following information pertains to United Ways, a private voluntary health and welfare organization, for the year ended December 31, 20X3. Balances in net assets at January 1, 20X3: Unrestricted...
-
In what ways is business writing different from school essays and private messages?
-
Plaintiffs purchased stock warrants (rights to purchase) for blocks of Osborne Computer Corp., the manufacturer of the first mass-market portable personal computer. Because of inability to produce a...
-
Your brother, Jackson, was laid off from his job with a large and famous software company. He would like to sell his stock in the company and use the proceeds to start a restaurant. The stock is...
-
If f(x) = x + 3 and g(x) = x + 8x + 15. Determine an equation for y = f(x) * g(x)
-
The following model is that of aspartame, C 14 H 18 N 2 O 5 , known commercially under many names, including NutraSweet. Only the connections between atoms are shown; multiple bonds are not...
-
Fill in any unshared electrons that are missing from the following linebond structures: (a) H3C CH3 Dimethyl disulfide (b) H3C NH Acetamide (c) H3C Acetate ion
-
Explain why hydroxide ion catalyzes the reaction of piperidine with 2,4-dinitroanisole, but has no effect on the reaction of piperidine with 1-chloro-2,4-dinitrobenzene. piperidine
-
A linear elastic material with a length of 36.0 mm and a square cross-section (side length = 2.3 mm) fails during tensile testing under a load of 76.3 N after being stretched to 40.7 mm. a. Calculate...
-
Matador Inc. owns 60% of Tital Co.s' outstanding common stock. Matador's liabilities total $350000, and Titan's liabilities total $200000. Included in Titan's financila statements is a $100000 note...
-
SELCO social enterprise provides sustainable energy solutions to low-income households and small businesses. In one scheme, solar-powered lighting was provided by SELCO to a silkworm farmer who...
-
Solve the differential equation with the given initial condition. 1x dx withx(10) = 2 dy 2 y 2y '
-
A car initially traveling at 31.2 m/s undergoes a constant negative acceleration of magnitude 1.40 m/s after its brakes are applied. (a) How many revolutions does each tire make before the car comes...
-
Using detailed cash-flow information, a financial analyst claims to be able to spot companies that are likely candidates for bankruptcy. The analyst is presented with information on the past records...
-
KD Insurance Company specializes in term life insurance contracts. Cash collection experience shows that 20 percent of billed premiums are collected in the month before they are due, 60 percent are...
-
a. Calculate the ratios of the different kinds of protons in a compound with an integral ratio of 6 : 4 : 18.4 (going from left to right across the spectrum). b. Determine the structure of a compound...
-
The two hydrogens of a methylene group adjacent to an asymmetric carbon are not equivalent hydrogens because they are in different environments due to the asymmetric carbon. (You can verify this...
-
In a reaction called the Birch reduction, benzene can be partially reduced to 1,4-cyclohexadiene by an alkali metal (Na, Li, or K) in liquid ammonia and a low-molecular-weight alcohol. Propose a...
-
A bar magnet is oriented with its magnetic dipole moment at 54.9 to the direction of a 0.0467 T magnetic field, causing a torque of 0.00227 Nm to act on it. Find the magnitude of the magnet's...
-
9. Three point charges are oriented as shown below: q = 40 C at the origin and q = -20 on the x-axis at x = 2, and 93 = electric force on q2 due the other charges? * 30c +93 30 C located at x = 2 cm,...
-
a) Determine the rate on the FRA(6x9) contract if the 6-month and 9-month LIBOR rates are 2.00% and 2.10%, respectively. Use day-counts of 182 and 272 for the two LIBOR rates b) Determine the dollar...

Study smarter with the SolutionInn App


