How can the following compounds be prepared, starting with a carbonyl compound with one fewer carbon atoms
Question:
How can the following compounds be prepared, starting with a carbonyl compound with one fewer carbon atoms than the desired product?
a. HOCH2CH2NH2
b.
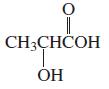
Transcribed Image Text:
CH3CHČOH ОН
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 54% (11 reviews)
A this can be prepared by the reaction of methyl c...View the full answer

Answered By

Raju Mandal
From my childhood , i was good in studying. I have completed my 10th exam from sagarpara high school and securing 1st in my batch.Then i have complete 12th standard from krishnath college school.i have completed my BSc in chemistry from krishnath college and my rank was 2nd in my college and 5th in kalyani university.Now i aam studying Master in school of chemistry , university of hyderabad
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
How could the following compounds be synthesized, starting with a hydrocarbon that has the same number of carbon atoms as the desired product? a. b. CH3CH2CH2CH2OH c. d. CH3CH2CH2CH2CH2Br CH...
-
How could the following compounds be prepared from a carbonyl compound with no carbon-carbon double bonds? a. b. CH CH-CHCCH2CH2CH3 C CH-CH2 CH3
-
Show how the following compounds could be prepared, starting with 3 cyanocyclohexanone a. b. CH2CH2CCH
-
Louise Kalbe drew a check in the amount of $7,260 payable to the order of cash on her account at the Pulaski State Bank. The check was lost or stolen, but Kalbe did not report this to the bank, nor...
-
Explain how signal amplification is achieved in enzymelinked immunosorbent assays.
-
Lewis is not an active participant in three rental activities, which have turned profits in recent years. The following gain and losses apply to the current year for Lewis: Gain or (Loss) Rental...
-
What is the difference between bivariate regression and multiple regression?
-
Dan Simms is the president and sole shareholder of Simms Corporation, 1121 Madison Street, Seattle, WA 98121. Dan plans for the corporation to make a charitable contribution to the University of...
-
choose two companies that are successfully following a Market Orientation (as described in the textbook) in 2023 and report on their market orientation execution. How does these companies take on a...
-
In a transposition cipher the encryption and decryption keys are often represented as two one-dimension tables (arrays) and the cipher is represented as a piece of software (a program). a. Show the...
-
Name the following compounds: a. b. c. CH;CHCH,CH,CH,CH3 OH
-
Explain why the aldehydic hydrogen (the one attached to the carbonyl carbon) is not exchanged with deuterium. -OD CH;CH CD;CH D20
-
Three identical books are stacked vertically. The normal force of book 1 on book 2 is A. Equal to the weight of one book. B. Less than the weight of one book. C. Greater than the weight of one book....
-
What is test checking? In what circumstances test checking is advisable? What factors are to be considered before resorting to test checking? What are the advantages and disadvantages of test...
-
Discuss various classes of error and state in each case the effect they might have on the trial balance being discovered.
-
Write short notes on: a. Batch total b. Test data c. Check digit
-
Discuss the advantages and disadvantages of balance sheet audit. Also state the auditors position in relation to balance sheet audit.
-
Distinguish between internal control system and internal check system.
-
What is the reasoning behind the fact that an infinite stream of cash flows has a finite present value?
-
What recommendations would you make to Big Four firms to help them (1) avoid confrontations with governmental officials in an authoritarian society and (2) deal effectively with such confrontations...
-
Two isomers of 1,2-dichloroethene are known. One has a dipole moment of 2.4 D; the other has zero dipole moment. Draw the two isomers and explain why one has zero dipole moment. CHCl=CHCl 1,...
-
Draw the hydrogen bonding that takes place between: (a) Two molecules of ethanol. (b) Two molecules of propylamine. (c) A molecule of dimethyl ether and two molecules of water. (d) Two molecules of...
-
For each pair of compounds, circle the compound you expect to have the higher boiling point. Explain your reasoning. (a) (CH3)3C-C(CH3)3 and (CH3)2CH-CH2CH2-CH(CH3)2 (b) CH3(CH2)6CH3 and...
-
Why was Thomas Paine's "common sense" article significant to the american revolution?
-
Aramex, Inc is currently offering a bond with a par value of $1,000, which is priced at $1,488 in the market. The bond offers a fixed annual coupon rate of 14.05 percent, which is paid semiannually....
-
The cash flow for the firm's project is -$40 million in year 0 and $19 million in years 1-4. After year 4, the FCF is expected to grow at a constant rate of 0.020. The firm's discount rate is 0.068....

Study smarter with the SolutionInn App


