How could you prepare hexan-2-one from the following starting materials? (a) (c) OH CH3CHCHCHCHCH3 CH3 CH3CHCHCHC=CH (b)
Question:
How could you prepare hexan-2-one from the following starting materials?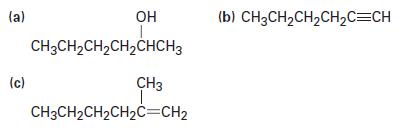
Transcribed Image Text:
(a) (c) OH CH3CH₂CH₂CH₂CHCH3 CH3 CH3CH₂CH₂CH₂C=CH₂ (b) CH3CH₂CH₂CH₂C=CH
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (4 reviews)
To prepare hexan2one from the given starting materials we need to perform a reaction that introduces ...View the full answer

Answered By

Collins Njuguna
I graduated from Maseno University with a Bachelor of Science in Applied Statistics. After graduation, I started tutoring students in mathematics. My experience in mathematics education is extensive and varied. I have taught a wide range of topics, including algebra, geometry, trigonometry, calculus, statistics, probability, and computer science. I have also worked with students of all ages and backgrounds, from elementary school to college.
My teaching method is based on the idea of hands-on learning. I believe that students learn best when they are actively engaged in the learning process, so I focus on giving students the tools they need to explore the material on their own. I also emphasize the importance of practice and review, as these are essential for mastering math concepts.
I have also developed several online and in-person courses on mathematics. My courses are designed to help students learn mathematics in an efficient and comprehensive way, and I use a variety of activities and exercises to ensure that my students are engaged and motivated.
Overall, my passion for mathematics and teaching has allowed me to be a successful tutor and educator in the field. I am confident that my experience will help your students master the mathematics they need to succeed.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
How could you prepare the following compounds with benzene as one of the starting materials? (a) (b)
-
How could you prepare the following compounds using a starting material containing no more than three carbon atoms? a. b. CH3 CH CH CHCHCH O Br CH CH2CH2CCH3
-
Starting with methyl propanoate, how could you prepare 4-methyl-3-heptanone? CH3CH3COCH3CH3CH CCHCH CH2CH3 methyl propanoate CH3 4-methyl-3-heptanone
-
Let (x) = x 2 + 3x + 2 and g(x) = x + 1. Find each of the following. (a) (fg)(x) (b) (fg) (-2) (c) (+)(x) (d) ()(-2)
-
You are the manager in a market composed of eight firms, each of which has a 12.5 percent market share. In addition, each firm has a strong financial position and is located within a 100- mile radius...
-
What is the bandwidth of the composite signal shown in Figure 3.37? Frequency 180 5 5 5 5
-
Many single women and married couples use donated sperm to conceive children each year. Pennsylvania resident Donna Donovan decided to use donated sperm from Idant Laboratories, a New York sperm bank...
-
Cost data for Tiwana Manufacturing Company for the month ended May 31, 2014, are as follows: a. Prepare a cost of goods manufactured statement for May 2014. b. Determine the cost of goods sold for...
-
Consider a 100-step binomial model where the price of a non-dividend-paying asset at time n, Sn is modelled as Sn = SoZ1 Z2... Zn where Z are i.i.d. random variables with u = 1.05 w. p. p Zi = d =...
-
How might you use a Grignard reaction of an aldehyde or ketone to prepare the following molecule (red=O)?
-
Predict the products of the reaction of the following substances with CrO 3 in aqueous acid: (a) CH3CHCHCHCH (b) CH3 CH3CH,CH,CH, CHO CH3 (c)
-
The circuit in Figure P8-17 is operating in the sinusoidal steady state with \(\omega=10 \mathrm{krad} / \mathrm{s}\). (a) What is the equivalent impedance of the circuit? (b) If one wanted to cancel...
-
You are currently only invested in the Natasha Fund (aside from risk-free securities). It has an expected return of 15% with a volatility of 19%. Currently, the risk-free rate of interest is 3.2%....
-
A big pharmaceutical company, DRIg, has just announced a potential cure for cancer. The stock price increased from $1 to $137 in one day. A friend calls to tell you that he owns DRIg. You proudly...
-
Your firm is planning to invest in an automated packaging plant. Harburtin Industries is an all-equity firm that specializes in this business. Suppose Harburtins equity beta is 0.88, the risk-free...
-
Four men and four women are shipwrecked on a tropical island. How many ways can they (a)Form four malefemale couples, (b)Get married if we keep track of the order in which the weddings occur,...
-
In mid-2009, Rite Aid had CCC-rated, 11-year bonds outstanding with a yield to maturity of 17.3%. At the time, similar maturity Treasuries had a yield of 2%. Suppose the market risk premium is 4% and...
-
Why are databases important for accounting information systems? Describe some concerns, and explain why each one is important.
-
Let (X. A. p) be a measure space. Show that for any A,B A, we have the equality: (AUB)+(An B) = (A) + (B).
-
How many signals would you expect in the 1 H NMR spectrum of each of the following compounds: (a) (b) (c) (d) (e) (f) CI Br -
-
How many signals would you expect in the 13 C NMR spectrum of each of the compounds in Problem 16.34? In Problem 16.34 How many signals would you expect in the 1 H NMR spectrum of each of the...
-
How would you distinguish between the following compounds using 13 C NMR spectroscopy?
-
What experiences did you have in your early learning years to support diverse cultures in the classroom? Do you feel there was enough exposure to diverse cultures? If not, how can you ensure you...
-
4. Tania is a fashion designer her late 30s that plans to retire within 20 years. She enjoys a very "in" lifestyle and likes to hang out with the "in-crowd" in the "in" nightspots whenever she has...
-
"Multiple IRRs. The Fairmont Corporation is planning on erecting an exhibit for the next state fair. The exhibit will require a cash outlay of $100,000 at the first of this year. Expected gate...

Study smarter with the SolutionInn App


