Predict the products of the reaction of the following substances with CrO 3 in aqueous acid: (a)
Question:
Predict the products of the reaction of the following substances with CrO3 in aqueous acid: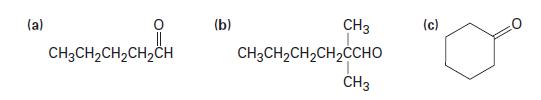
Transcribed Image Text:
(a) CH3CH₂CH₂CH₂CH (b) CH3 CH3CH,CH,CH, CHO CH3 (c)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 60% (5 reviews)
To predict the products of the reactions with CrO3 in aqueous acid we need to consider the type of r...View the full answer

Answered By

User l_1006857
I am a computer science professional with expertise in databases, AI programming, data structures and algorithms, and mathematics. With a strong background in these areas, I possess the knowledge and skills necessary to design and optimize database systems, develop intelligent algorithms and models, and solve complex computational problems. My proficiency in SQL, NoSQL, machine learning techniques, and mathematical concepts equips me to contribute to innovative projects and drive technological advancements.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
What products would you expect from oxidation of the following compounds with CrO3 in aqueous acid with pyridinium chlorochromate? (a) 1-Hexanol (b) 2-Hexanol (c) Hexanol
-
Predict the products of the following acid-base reactions. Using pKa values, indicate which side of each equilibrium reaction is favored, and label the species representing the stronger acid and...
-
Name the following alkenes, and predict the products of their reaction with (1) meta-chloroperoxybenzoic acid, (ii) KMnO4 in aqueous acid, and (iii) O3, followed by Zn in aceticacid: (a) (b)
-
For each polynomial function, find (a) (-1), (b) (2), and (c) (0). f(x) = x + 2x - 8
-
Barnacle Industries was awarded a patent over 15 years ago for a unique industrial- strength cleaner that removes barnacles and other particles from the hulls of ships. Thanks to its monopoly...
-
Why do optical signals used in fiber optic cables have a very short wave length?
-
Panther Racing, LLC, is involved in competitive racing, including the development, maintenance, and operation of a racing team in the IndyCar series. Beginning in 2008, the National Guard annually...
-
Cheryl Montoya picked up the phone and called her boss, Wes Chan, the vice president of marketing at Piedmont Fasteners Corporation: Wes, Im not sure how to go about answering the questions that came...
-
-. Use the Average Annual Costs of Owning and Operating a Car table below. If you drive 40,000 miles per year, what is the total annual expense for an electric car? (2 points) Average Annual Costs of...
-
How could you prepare hexan-2-one from the following starting materials? (a) (c) OH CH3CHCHCHCHCH3 CH3 CH3CHCHCHC=CH (b) CH3CHCHCHC=CH
-
Propose structures for molecules that meet the following descriptions: (a) A ketone, C 5 H 10O (b) An aldehyde, C 6 H 10O (c) A keto aldehyde, C 6 H 10 O 2 (d) A cyclic ketone, C 5 H 8 O (a) (d)...
-
Based on the following data, estimate the model: (1/Yi) = B1 + B2Xi + ui a. What is the interpretation of B2? b. What is the rate of change of y with respect to X? c. What is the elasticity of y with...
-
In mid-2015, Cisco Systems had a market capitalization of $99 billion. It had A-rated debt of $18 billion as well as cash and short-term investments of $52 billion, and its estimated equity beta at...
-
Charlie draws 5 cards out of a deck of 52. If he gets at least three of one suit, he discards the cards not of that suit and then draws until he again has 5 cards. For example, if he gets 3 hearts, 1...
-
Weston Enterprises is an all-equity firm with two divisions. The soft drink division has an asset beta of 0.53, expects to generate free cash flow of $76 million this year, and anticipates a 4%...
-
John takes the bus with probability 0.3 and the subway with probability 0.7. He is late 40% of the time when he takes the bus, but only 20% of the time when he takes the subway. What is the...
-
In a group of five items, two are defective. Find the distribution of N, the number of draws we need to find the first defective item. Find the mean and variance of N.
-
What is prototyping? Under what circumstances should prototyping be used? Under what circumstances should it not be used?
-
Extend Algorithms 3.4 and 3.5 to include as output the first and second derivatives of the spline at the nodes.
-
Predict the multiplicity of each signal in the 1 H NMR spectrum of the following compound:
-
For each pair of compounds, identify how you would distinguish them using either 1 H NMR spectroscopy or 13 C NMR spectroscopy: (a) (b) (c) (d) 'CI CI CI
-
A compound with molecular formula C 8 H 18 exhibits a 1 H NMR spectrum with only one signal. How many signals would you expect in the 13 C NMR spectrum of this compound?
-
One example of a direct exchange quote is Euro 0.82 per $ in the U.S. O True False
-
In reading the Apology, we drew a major distinction between the passional motivations in the human soul (the Psyche') and a rational motive. In the chariot analogy for the soul, what two particular...
-
Calculation of compound interest A sum of $1000 is invested at an interest rate of 12% annually. Calculate the amounts in the account after three years if interest is compounded annually,...

Study smarter with the SolutionInn App


