How many signals would you expect to see in the 1 H NMR spectrum of each of
Question:
How many signals would you expect to see in the 1H NMR spectrum of each of the following compounds?
a. CH3CH2CH2CH3
b. BrCH2CH2Br
c. CH2=CHCl
d.
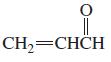
e.
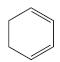
f.
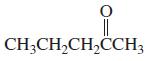
g.
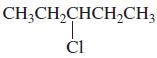
h.
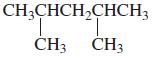
i.
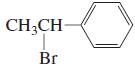
j.
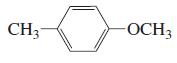
k.
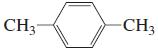
l.
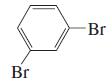
m.
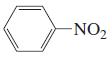
n.
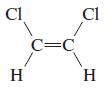
o.
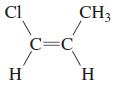
Transcribed Image Text:
CH2=CHCH
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 77% (9 reviews)
a 2 signals 6 hydrogen on terminal CH3 group forms a signal and the hydrogen of CH2 grou...View the full answer

Answered By

DIPIN DAS AK
I completed my masters in chemistry from CUSAT, kerala, India. I studied bachelor of science from Calicut University and i had a project in nanotechnology from Hyderabad central university. I have two year teaching experience in higher secondary level in India as a private tutor. Now i work as a subject matter expert in conects app for part time. I love teaching and sharing knowledge.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
How many signals would you expect in the 1 H NMR spectrum of each of the following compounds: (a) (b) (c) (d) (e) (f) CI Br -
-
How many signals would you expect to find in the 1H NMR spectrum of each of the following compounds? (a) 1-Butanol (b) Butane (c) 1,4-Dibromobutane
-
How many 1H NMR signals would you expect from each of the following compounds? (a) (b) (c) (d) (e) (f) (g) (h) (i) (j) (k) (l) (m) (n) (o) (p) OH CI
-
What other cost factors might you include in such an economic analysis? The proposed small office building in Example 3-2 has 24,000 net square feet of area heated by a natural gas furnace. The owner...
-
Light passes from benzene (medium 1) to water (medium 2) in Figure 19-19 at (a) 1 = 30 or (b) 1= 0. Find the angle 2 in each case.
-
Marvin Johnson is an Environmental Engineer for one of several local plants whose water discharges flow into a lake in a flourishing tourist area. Included in Marvins responsibilities is the...
-
Help for Insomniacs A recent study shows that just one session of cognitive behavioral therapy can help people with insomnia. In the study, forty people who had been diagnosed with insomnia were...
-
Jefferson Products, Inc., is considering purchasing a new automatic press brake, which costs $300,000 including installation and shipping. The machine is expected to generate net cash inflows of...
-
Excerpts from Andre Company's December 31, 2024 and 2023, financial statements are presented below: Accounts receivable Inventory Net sales Cost of goods sold Total assets Net income Total...
-
Please complete the Express Catering, Inc.s 2014 tax return based upon the information provided below. If required information is missing, use reasonable assumptions to fill in the gaps. Ignore any...
-
Which of the following compounds has a vibration that is infrared inactive: acetone, 1-butyne, 2-butyne, H 2 , H 2 O, Cl 2 , ethene?
-
How could you distinguish the 1 H NMR spectra of the following compounds? a. CH 3 OCH 2 OCH 3 b. CH 3 OCH 3 c. CH3 CH3OCH,CCH2OCH3 H3
-
How does the expense warranty approach differ from the sales warranty approach?
-
This case is about the social network Facebook, which hardly needs an introduction. However, when we first featured it in an earlier edition of his text, it was a niche service mainly used in...
-
Explain the options for applying social media marketing to B2B marketing campaigns.
-
What is website prototyping? Give three benefits of this approach.
-
Explain personalisation in a digital marketing context.
-
How should companies decide on the granularity of targeting in digital media campaigns? Select two digital media channels to illustrate your examples.
-
How are the periodic coupon payments on a bond determined?
-
A bar of length = 1 has one fixed and one free end and stiffness function c(x) = 1 - x. Find the displacement when subjected to a unit force. Pay careful attention to the boundary condition at the...
-
(a) What type of pericyclic reaction is required to form benzene from Dewar benzene? (b) Explain why Dewar benzene, although a very un-stable molecule, is not readily transformed to benzene....
-
Show both conrotatory processes for the thermal electtrocyclec conversion of (2E, 4E) - 2,4 -hexadiene into 3,4- dimethylcyclobutene (Eq. 27.8 ). Explain why the two processes are equally likely.
-
Show both conrotatory processes for the thermal electtrocyclec conversion of (2E, 4E) - 2,4 -hexadiene into 3,4- dimethylcyclobutene (Eq. 27.8 ). Explain why the two processes are equally likely.
-
Can you elucidate the intricacies of cellular respiration, delineating the metabolic pathways involved and their respective roles in energy production within eukaryotic organisms ?
-
How do the mechanisms of ventilation and gas exchange operate synergistically in facilitating the diffusion of oxygen and carbon dioxide across the respiratory membrane, ensuring optimal...
-
How do environmental factors, such as altitude, temperature, and atmospheric composition, influence respiratory physiology, necessitating adaptive responses at both the cellular and systemic levels...

Study smarter with the SolutionInn App


