How could you distinguish the 1 H NMR spectra of the following compounds? a. CH 3 OCH
Question:
How could you distinguish the 1H NMR spectra of the following compounds?
a. CH3OCH2OCH3
b. CH3OCH3
c.
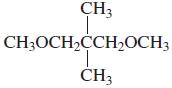
Transcribed Image Text:
CH3 CH3OCH,CCH2OCH3 ČH3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 60% (10 reviews)
CH3OCHOCH3...View the full answer

Answered By

Shubham Belsare
I'm currently pursuing Post graduation in organic chemistry and I'm passionately driven to teach Organic Chemistry to the students who finds hard time learning the subject. Apart from my regular college, I'm also a part-time subject expert on Chegg and I very much like and enjoy clearing the doubts of students which also increase my knowledge toward the subject.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
How would integration distinguish the 1H NMR spectra of the following compounds? CH2Br CH3 CHj CH2Br Br CH3
-
How could 1H NMR spectra distinguish the following compounds? NO2 NO2 Br
-
How could you distinguish among the following compounds, using a. Their infrared spectra? b. Their 1H NMR spectra? CH2OH CH OH CH2OCH3 CH COCH3 CCH3
-
The following items are dropped from an airplane. Rank them in order from lowest terminal speed to highest and justify your ranking. (a) Bowling ball (b) Beach ball (c) Spear or javelin (pointing...
-
In the cavity ring-down measurement at the opening of this chapter, absorbance is given by where L is the length of the cavity between mirrors, c is the speed of light,t is the ring down lifetime...
-
Suppose you were going to design a front door and doorway to fit snugly enough to keep out the drafts, yet to be easy to open. (You are not showing off precision carpentry here, but merely designing...
-
Home Realty, Incorporated, has been operating for three years and is owned by three investors. J. Doe owns 60 percent of the total outstanding stock of 9,000 shares and is the managing executive in...
-
Terry Kunkle and VanBuren High hosted a Christmas party in Berkeley County, South Carolina. Guests had drinks and hors doeuvres at a residence and adjourned to dinner in a barn across a public road....
-
Supervision of the Audit Engagement Of the standard, paragraph . 0 6 states the engagement partner and other engagement team members performing supervisory activities should take into account: State...
-
Sixty-three percent of U.S. adults oppose special taxes on junk food and soda. You randomly select 10 U.S. adults. Find the probability that the number of U.S. adults who oppose special taxes on junk...
-
How many signals would you expect to see in the 1 H NMR spectrum of each of the following compounds? a. CH 3 CH 2 CH 2 CH 3 b. BrCH 2 CH 2 Br c. CH 2 =CHCl d. e. f. g. h. i. j. k. l. m. n. o. CH2=CHCH
-
Where would you expect to find the 1 H NMR signal of (CH 3 ) 2 Mg relative to the TMS signal? (See Table 12.3.) Table 12.3 The Electronegativities of Some of the Elementsa IA IIA IB IIB IVA VA VIA...
-
The Motuto Equipment Corporation maintains a general ledger account for each class of inventory, debiting the individual accounts for increases during the period and crediting them for decreases. The...
-
How can online PR help to promote a new product?
-
Name, and briefly explain, four characteristics of an online service that will govern whether a user recommends it.
-
Suggest three measures a company can take to ensure that a customers privacy is not infringed when conducting one-to-one marketing.
-
Explain two applications of dynamic pricing on the Internet.
-
How can different forms of customer insight be used to inform campaign execution?
-
How will the yield to maturity of a bond vary with the bond's risk of default?
-
Show that if A is any m n matrix, then Im A = A and AIn = A.
-
After heating to 200 °C, the following compound is converted 95% yield into an isomer A that can be hydrogenated o cyclodecane. Give the structure of A, including its stereo chemistry.
-
After heating to 200 °C, the following compound is converted 95% yield into an isomer A that can be hydrogenated o cyclodecane. Give the structure of A, including its stereo chemistry.
-
(a) Nitrogen has relatively stable isotopes (half-life greater than 1 second) of mass numbers 13, 14, 15, 16, and 17. (All except 14N and 15N are radioactive.) Calculate how many protons and neutrons...
-
Reflecting on the book Who Gets Promoted, Who Doesn't, and Why (Asher, 2008), answer the following questions in depth. Explain the statement, "all business is sales." What six things matter? If...
-
Reflecting on the book Who Gets Promoted, Who Doesn't, and Why (Asher, 2008), answer the following questions in depth. No matter what you have done in the past, the boss really doesn't care. Why?...
-
Do you think Richard Scrushy should have been found guilty or acquitted for his role in the case against HealthSouth? https://www.wsj.com/articles/SB111702610398942860

Study smarter with the SolutionInn App


