How might you prepare the following amines from ammonia and any alkyl halides needed? (a) CH3CHCHCHCHCHNH CHNH2
Question:
How might you prepare the following amines from ammonia and any alkyl halides needed?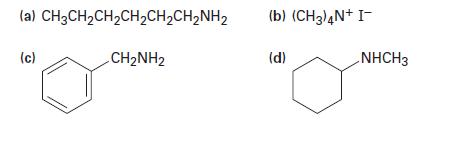
Transcribed Image Text:
(a) CH3CH₂CH₂CH₂CH₂CH₂NH₂ CH₂NH2 (c) (b) (CH3)4N+ I- (d) NHCH3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 42% (7 reviews)
To prepare the given amines from ammonia NH3 and the required alkyl halides we can use a simple nucl...View the full answer

Answered By

Deepak Sharma
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
How could you prepare the following amines from ammonia and appropriate alkyl halides? (a) Triethylamine (b) Tetramethylammonium bromide
-
How might you prepare each of the amines in Problem 12.32 from butan-1-ol? Problem 12.32 How might you prepare the following amines from 1-bromobutane? (a) Butylamine (b) Dibutyl amine (c) Pentylamine
-
How might you prepare the following amines from 1-bromobutane? (a) Butylamine (b) Dibutyl amine (c) Pentylamine
-
In Exercises 8586, find a. (f g)(x); b. (g f)(x); c. (f g)(3). f(x) = x 2 + 3, g(x) = 4x - 1
-
Franks Fun Company owns 30 pizza parlors and a minor league baseball team. During the current year, the company sold three of its pizza parlors and closed another when the lease on the building...
-
The indented parenthetic representation of a tree T is a variation of the parenthetic representation of T (see Code Fragment 8.26) that uses indentation and line breaks as illustrated in Figure 8.22....
-
Crush Autosmashers can purchase a new electromagnet for moving cars at a cost of \($20,000.\) At the end of its useful life, the electromagnet will be worth \($1,000.\) If Crushs MARR is 12...
-
Fleming, chief administrator for Valley View Hospital, is concerned about the costs for tests in the hospitals lab. Charges for lab tests are consistently higher at Valley View than at other...
-
19. Write your first derivative of the following function in the space on the answer sheet. Y 2X2+5X3-X+10 20. Write your first derivative of the following function in the space on the answer sheet....
-
Show the products of the following reactions: (a) (c) CH3CHCHNH CH3CH2CNH2 CH3Br 1. LiAlH4 2. HO ? ? (b) (d) NH CEN HBr ? 1. LIAIH4 2. HO ?
-
Draw structures of compounds that meet the following descriptions: (a) A secondary amine with one isopropyl group. (b) A tertiary amine with one phenyl group and one ethyl group. (c) A quaternary...
-
The statement of cash flows for Riker Company (prepared using the indirect method) follows. Consider the following additional information: (a) Sales for the year totaled $812,350. Cost of goods sold...
-
You are the primary care physician of Margo, the 50-year- old woman who has dementia and has developed pneumonia. Do you overrule her advance directive and give her the antibiotics, or do you follow...
-
Think about a time you experienced an especially effective example of entertainment education. What features of your example (e.g., messages, characters, drama) made it so effective? How could those...
-
What health- related issue do you perceive as the most in need of awareness? What type of campaign/intervention style would you use to increase awareness? Why would your strategy be effective?
-
Campaign researchers are looking to Internet- based technologies as the future of campaigns and interventions. Ten years from now, how do you expect the Internet will be utilized in campaign and...
-
The CDC, FEMA, and DHS have all used the zombie apocalypse to promote emergency preparedness. Do you think this attention- getting tactic minimizes the importance of emergency preparedness planning?...
-
Multiple Choice Questions 1. Which of the following funds can be used to account for the spendable income from a Private Purpose Trust Fund? a. Agency Fund b. General Fund c. Capital Projects Fund d....
-
The following information is for Montreal Gloves Inc. for the year 2020: Manufacturing costs Number of gloves manufactured Beginning inventory $ 3,016,700 311,000 pairs 0 pairs Sales in 2020 were...
-
Using acetylene as your only source of carbon atoms, design a synthesis of pen-tanal. (Pentanal has an odd number of carbon atoms, while acetylene has an even number of carbon atoms): H.
-
The regions of + in a compound are the regions most likely to be attacked by an anion, such as hydroxide (HO - ). In the compound below, identify the two carbon atoms that are most likely to be...
-
Consider the three compounds shown below and then answer the questions that follow: a) Which two compounds are constitutional isomers? b) Which compound contains a nitrogen atom with trigonal...
-
Port 26. BOATING The light from a lighthouse can be seen from an 18-mile radius. A boat is anchored so that it can just see the light from the lighthouse. A second boat is located 25 miles from the...
-
For the beam illustrated in the figure, find the locations and magnitudes of the maximum tensile bending stress due to Mand the maximum shear stress due to V. Parameters are a = 310 mm, b = 160 mm, c...
-
(Related to Checkpoint 5.2) (Future value) Leslie Mosallam, who recently sold her Porsche, placed $10,400 in a savings account paying annual compound interest of 5 percent. a. Calculate the amount of...

Study smarter with the SolutionInn App


