How would you prepare benzylamine, C 6 H 5 CH 2 NH 2 , from each of
Question:
How would you prepare benzylamine, C6H5CH2NH2, from each of the following starting materials?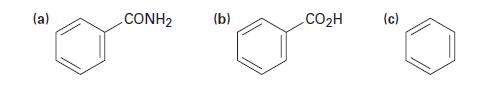
Transcribed Image Text:
(a) CONH2 (b) CO₂H (c)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (4 reviews)
To prepare benzylamine C6H5CH2NH2 from each of the given starting materials we need to perform speci...View the full answer

Answered By

Deepak Sharma
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Show how you might prepare benzylamine from each of the following compounds: (a) (b) (c) Benzyl bromide (two ways) (d) Benzyl tosylate (e) Benzaldehyde (f) Phenylnitromethane (g) NH2 Benzylamine CN...
-
How would you prepare the following compounds from the given starting materials? a. b. c. d. CH3CH2CH CH3CHCH N(CH3)2 CH CHCH OCH3
-
Planning is one of the most important management functions in any business. A front office managers first step in planning should involve determine the departments goals. Planning also includes...
-
In Exercises 6567, consider a scalar function and a vector field F in space. Determine whether the expression is a vector field, a scalar function, or neither. Explain. div[curl()]
-
List and describe four important differences between managerial and financial accounting.
-
Describe a method for finding the middle node of a doubly linked list with header and trailer sentinels by link hopping, and without relying on explicit knowledge of the size of the list. In the case...
-
If a well-behaved investment alternative's internal rate of return (IRR) is equal to MARR, which of the following statements about the other measures of worth for this alternative must be true? 1....
-
The following project is at the end of its sixth week. Find the cost and schedule variances. Also find the CPI and SPI. Then find the critical ratio of the project using earned value calculations....
-
Supposons un consommateur ayant une richesse W qui est distribue selon une loi de densit de probabilit fw (w). Montrez qu'on peut obtenir une approximation du cot du risque, CR, en utilisant: 1 CRrr...
-
Which compound is more basic, triethylamine or aniline? Does the following reaction proceed as written? (CH3CH)2NH+ CI + NH NH3+ CI + (CH3CH2)2N
-
How might you prepare the following amines from 1-bromobutane? (a) Butylamine (b) Dibutyl amine (c) Pentylamine
-
Gordon Hammock Company produces a variety of hammocks and other outdoor products. The company uses job costing and applies overhead to work in process using a predetermined overhead rate, with direct...
-
The FDA issued a final compliance guidance related to pharmacy compounding. In the guidance, the FDA clarified which activities compounding pharmacies could lawfully engage and which activities the...
-
You are the only pharmacist at a meeting with other healthcare professionals. A physician brings up the topic of DTC drug ads on television and in magazines, lamenting that the ads are so seductive...
-
A patient asks the pharmacist whether the FDA regulates cosmetics and if so, in what manner. How should the pharmacist answer the patient?
-
A patient purchasing syringes and needles for insulin injection asked the pharmacist whether the FDA regulates these products and if so, in what manner. Provide a complete response to this patient.
-
Which of the following are not examples of a vicious cycle of deleveraging? Explain. a. Your university decides to sell several commercial buildings in the middle of town in order to upgrade...
-
Obtain the CAFR of a governmental unit, and read the MD& A. Describe three significant comments made by management.
-
The landing gear of an aircraft with: mass of 2000 kg the spring-mass-damper system Consider that the runway surface is y(t) = 0.2 cos 157.08t stiffness of the spring is 5 x 105 N/m. What is the...
-
Propose a plausible synthesis for each of the following transformations: a. b. c. Br Br
-
There are four constitutional isomers with molecular formula C 3 H 9 N. Draw a Lewis structure for each isomer and determine the number of lone pairs on the nitrogen atom in each case?
-
Would water be a suitable proton source to protonate the following compound? ONa
-
deposit 50000, APY is 5.05%, how much can I get after a year?
-
Consumer loans are: Question options: Long-term loans, which are typically paid on an installment basis Short-term loans that are usually single-payment loans Bank credit cards All of the above are...
-
Linda recently invested in real estate with the intention of selling the property one year from today. She has modeled the returns on that investment based on three economic scenarios. She believes...

Study smarter with the SolutionInn App


