Question: Would water be a suitable proton source to protonate the following compound? ONa
Would water be a suitable proton source to protonate the following compound?
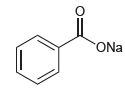
ONa
Step by Step Solution
★★★★★
3.33 Rating (165 Votes )
There are 3 Steps involved in it
1 Expert Approved Answer
Step: 1 Unlock

No water would not be a suitable proto... View full answer

Question Has Been Solved by an Expert!
Get step-by-step solutions from verified subject matter experts
Step: 2 Unlock
Step: 3 Unlock


