Name the following ketones and aldehydes. When possible, give both a common name and an IUPAC name.
Question:
Name the following ketones and aldehydes. When possible, give both a common name and an IUPAC name.
a. CH3CO(CH2)4CH3
b. CH3(CH2)2CO(CH2)2CH3
c. CH3(CH2)5CHO
d. PhCOPh
e. CH3CH2CH2CHO
f. CH3COCH3
g. CH3CH2CHBrCH2CH(CH3)CHO
h. Ph-CH=CH-CHO
i. CH3CH=CH-CH=CH-CHO
j.
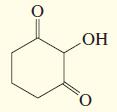
k.
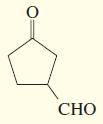
l.
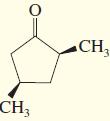
Transcribed Image Text:
HO
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (12 reviews)
a IUPAC NAME Heptan2one COMMON NAME methyl namyl ketone b IUPAC NAME 4Heptanone c IUPAC NAME H...View the full answer

Answered By

Rinku Pahel
I have given tuitions to 5-12 class. I have 4 years of experience in tuition.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Name the following carboxylic acids (when possible, give both a common name and a systematic name). (a) (b) (c) (d) (e) (f) CH3 CH2CH, COOH COOH NO NO. COOH COOH HO Cl COOH COOH
-
Name the following carboxylic acid derivatives, giving both a common name and an IUPAC name where possible. (a) PhCOOCH2CH(CH3)2 (b) PhOCHO (c) PhCH(CH3) COOCH3 (d) PhNHCOCH2CH(CH3)2 (e) CH3CONHCH2Ph...
-
Give a common name (when possible) and a systematic name for each compound. (a) CH3OCH = CH2 (b) CH3CH2OCH(CH3)2 (c) ClCH2CH2OCH3 (d) (e) (f) (g) (h) CH3C = CCH2OCH3 (i) OCH CH3 OCHj OCH3 " OCH3 OCH3
-
Find the derivative of the function. y = e x-4
-
Use Table 6-1 to calculate the pH of pure water at (a) 25C and (b) 100C.
-
Mozart Inc.s $98,000 taxable income for 20X1 will be taxed at the 21% corporate tax rate. For tax purposes, its depreciation expense exceeded the depreciation used for financial reporting purposes by...
-
In the decision-making process, what are the ways in which context can impact outcomes?
-
Multiple Choice Questions 1. Candy Corp. is a C corporation that began operations in year 1. Candy Corp.'s year 1 through year 3 taxable earnings and profits (E & P) are computed as follows. Year E &...
-
According to the Becker model, parents have children until the expected present value of the marginal benefits from having children equals the expected present value of marginal costs. Explain why...
-
Aquatic Line Company (ALC) manufactures a variety of strong and durable ropes. The company manufactures all their products in a large factory located near Nephi, Utah. All the types of ropes the...
-
Draw structures of the following derivatives. (a) The 2,4-dinitrophenylhydrazone of benzaldehyde (b) The semicarbazone of cyclobutanone (c) Cyclopropanone oxime (d) The ethylene acetal of hexan-3-one...
-
Predict the major products of the following reactions. a. b. c. d. e. f. g. h. i. j. PHNHNH2 H+
-
Leapin Larrys Pre-Owned Cars has two divisions, Operations and Financing. Operations is responsible for selling Larrys inventory as quickly as possible and purchasing cars for future sale. Financing...
-
Do you believe that your education has prepared you adequately for the writing you will do in your job? In what specific ways might that writing differ from the writing you did for your courses, both...
-
This chapter argues that our responses to communication situations tend to be generic (that is, patterned according to genres), and it describes the advantages of learning and working with the genres...
-
How transferable are genres? To what extent are genres specific to a particular workplace or professional community (e.g., Cell tech, an Atlanta-based cell-phone manufacturer) and to what extent are...
-
As a student, you have read and written documents in several different academic (and perhaps other) genres. When you enter the workplace, you will likely have to learn a new set of genres. Which...
-
How important do you believe it is to be familiar with the industry for which you will be writing? How might you acquire that familiarity? Also, do you think it will be more important to know about...
-
Selected liability items for Waugh Corporation at December 31, 2014, follow. Prepare the liabilities section of Waugh's balance sheet. Accounts payable Bonds payable, due 2028 $ 48,000 Income tax...
-
A 2500-lbm car moving at 15 mi/h is accelerated at a constant rate of 15 ft/s 2 up to a speed of 50 mi/h. Calculate force and total time required?
-
Using benzene and any other reagents, outline a synthesis of each of the following compounds. Tert-butylcyclohexane
-
Give the products expected (if any) when nitrobenzene reacts under the following conditions. (a) Cl2, FeCl3, heat (b) Fuming HNO3, H2SO4 (c) H3C-C-CI, AICI,( 1.1 equiv.), then H2O
-
Explain how you would distinguish between ethylbenzene, p-xylene, and styrene solely by NMR spectroscopy.
-
Question: Part One: Memo Write an analysis of the impact of tax laws and incentives as demonstrated in this current years tax return. Specifically, you must address the following: Explain the...
-
From the following ledger balances, prepare a trial balance for the Cheyenne Corp. at June 3 0 , 2 0 2 2 . All account balances are normal.Accounts Payable $ 1 0 , 0 0 0 , Cash $ 7 , 4 0 0 , Common...
-
Kathy is mixing fruit punch in a 32-cup punch bowl for a party. She plans to add at least 20 cups of fruit juice to the bowl before adding ginger ale. Identify the graph that represents the amounts...

Study smarter with the SolutionInn App


