Predict the major products of the following reactions. a. b. c. d. e. f. g. h. i.
Question:
Predict the major products of the following reactions.
a.
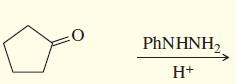
b.
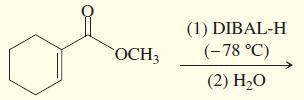
c.
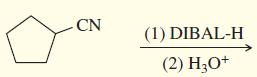
d.

e.
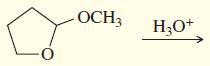
f.
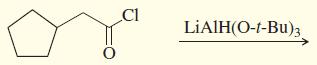
g.

h.
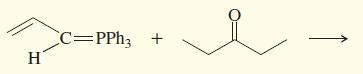
i.
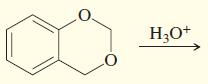
j.
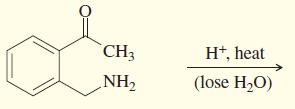
Transcribed Image Text:
PHNHNH2 H+
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 63% (11 reviews)
c d e f g h i j a b NHNHPh OCH3 PhNHNH H ...View the full answer

Answered By

Ujjwal Ghosh
I have been teaching for last 10 years. Many students whom I've taught are now studying the courses of their choice. Most of my students cracked the competitive examinations in the first attempt.
I've taught many students from graduation to higher level like master degree courses.
They were very happy with my teaching style.
I can solve higher order thinking questions in very less time.
https://www.linkedin.com/in/ujjwal-ghosh-1a506a1b4/
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Predict the products of the following reactions. (a) (b) (c) (d) (e) (f) (g) (h) (i) (j) (k) (l) OH CI CH NH2 Ph-C-CI+ NH2 0 + (D LiAIH (2) H20 (2) H,0 -. O (I) excess PhMgBr (2) H,o ( CH Mgl (2)...
-
Predict the products of the following reactions. (a) (b) (c) (d) (e) (f) (g) (h) CN A OMe O+ OMe A (2) NaBHs (3) H2O OMe MeO 0 CN CN
-
Predict the product of the following reactions. (a) (b) (c) (d) (e) (f) (g) (h) (i) HBr (excess), OH (1) NaH OH Br2 (excess) H20 H3C OH excess QH (1) NaH,Br (2) heat HNO3, H2SO4 NaNH2, NHa (1) NaOH...
-
Sketch the graph of the function. y = e -x/2
-
Calculate [H+] and pH for the following solutions: (a) 0.010 MHNO3 (b) 0.035 MKOH (c) 0.030 M HCl (d) 3.0 M HCl (e) 0.010 M [(CH3)4N+OH- Tetramethylammonium hydroxide
-
Kent Inc.s reconciliation between financial statement and taxable income for 20X1 follows: Pre-tax financial income ........................................ $150,000 Permanent difference...
-
It is mentioned that for a "decision to be made" there must be more than one alternative under consideration and the possible outcomes must be of unequal value. What are the three general types of...
-
Aztec Interiors deposits all cash receipts each Wednesday and Friday in a night depository, after banking hours. The data required to reconcile the bank statement as of May 31 have been taken from...
-
Suppose an individual's utility function over income (M) and leisure (L) is U(M,L)-L, and the individual has non-labor income Y and earns wage w. The total amount of time available is 16 hours. a....
-
Is a randomly generated 4-CNF sentence with n symbols and m clauses more or less likely to be solvable than a randomly generated 3-CNF sentence with n symbols and m clauses? Explain.
-
Name the following ketones and aldehydes. When possible, give both a common name and an IUPAC name. a. CH 3 CO(CH 2 )4CH 3 b. CH 3 (CH 2 ) 2 CO(CH 2 ) 2 CH 3 c. CH 3 (CH 2 ) 5 CHO d. PhCOPh e. CH 3...
-
Rank the following carbonyl compounds in order of increasing equilibrium constant for hydration: CH,COCH,CI CICH,CHO CH,0 CH3COCH3 CH3CHO
-
Yasukuni Corporation reported the following selected information in its general ledger at June 30: Accounts Receivable All sales were on account. Some accounts receivable were collected. One account...
-
Pick any technological object you use frequently: a cell phone, a laptop computer, an MP3 player. What different kinds of knowledge do you need to use the object? To use the object well? (Think about...
-
Technical communication jobs are considered writing jobs, yet this chapter talks about all kinds of user analysis, planning, project tracking and testing, and other activities. Are there any jobs...
-
How might the technology used in your professional workplace compare with or differ from the kinds of technology you learned in school and the ways you used it in school? How can you best prepare...
-
The summary for this chapter notes that genres are "shared conventions of text and situation." But it also notes that communicators have a great deal of flexibility in how they compose documents in...
-
More and more organizations are publishing their information online. Shouldn't technical communicators be able to reduce the production stage to simply saving something as a PDF and posting it to a...
-
Cooke Inc. issued a $240,000, 10-year, 8% note payable on October 1, 2013. The terms provide for blended payments of $8,773 payable in quarterly instalments on January 1, April 1, July 1, and October...
-
Write a paper about how diet relates to breast cancer in women study design to use: case control study purpose & rationale the purpose of this final project is to utilize the methods and...
-
Explain why The methyl group in the following compound has an unusual chemical shift of (- 1.61), about 4 ppm lower than the chemical shift of a typical allylic methyl group. : Na sodium salt of...
-
Explain why The methyl group in the following compound has an unusual chemical shift of (- 1.61), about 4 ppm lower than the chemical shift of a typical allylic methyl group. : Na sodium salt of...
-
Within each set, which compound should show NMR absorptions with the greater chemical shifts? Explain your choices. (1) (2)
-
E3-20 (Algo) Analyzing, Recording, and Evaluating the Effects of Income Statement Transactions [LO 3-3, LO 3-4, LO 3-5] [The following information applies to the questions displayed below.] MyBnB...
-
Required information [The following information applies to the questions displayed below.] The following data reports on the July production activities of the Molding department at Ash Company....
-
Question 1 Vieux Enterprise's total overhead costs at various levels of activity follow: Month Machine- Hours Total Overhead Costs April 70,000 $198,000 May 100,000 270,000 June 80,000 222,000 July...

Study smarter with the SolutionInn App


