Show how you could prepare the following substances from cyclohexanol: (a) (b) -Br (c) (d)
Question:
Show how you could prepare the following substances from cyclohexanol: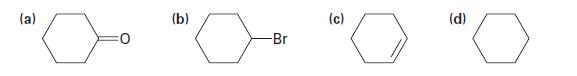
Transcribed Image Text:
(a) (b) -Br (c) (d)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (4 reviews)
To prepare the following substances from cyclohexanol we need to perform specific chemical reactions to transform cyclohexanol into the desired produc...View the full answer

Answered By

Saleem Abbas
Have worked in academic writing for an a years as my part-time job.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Show how you could prepare the following substances from propan-1-ol: (a) O || CH3CHCH (b) O CH3CHCOH (c) CH3CHCHO Na+ (d) CH3CHCHCl
-
Show how you could prepare each of the following compounds. Use the starting material indicated along with ethyl acetoacetate or diethyl malonate and any necessary inorganic reagents. Assume also...
-
Show how you could prepare each of the following compounds from cyclopentanone, D2O, and any necessary organic or inorganic reagents. H OH
-
Why is the number of equivalent units for materials only sometimes equal to the equivalent units for conversion?
-
A monopolists inverse demand function is P = 150 - 3Q. The company produces output at two facilities; the marginal cost of producing at facility 1 is MC1 (Q1) = 6Q1, and the marginal cost of...
-
Why do HTTP, SMTP, and POP3 run on top of TCP rather than on UDP?
-
XCBob, Inc. is an online motorcycle and ATV parts business from the owners home. XCBob entered into oral agreements with the defendant, Ed Tucker Distributers, for purchases of various materials to...
-
Lisa Kohl (age 44) is an unmarried high school principal. Lisa received the following tax documents: During the year, Lisa paid the following amounts (all of which can be substantiated): Home...
-
Suppose the 3-month interest rate (i) is 4%/year in New York and 8% in Frankfurt, and the $/ FD = 2%/year. A US investor would then invest in Frankfurt by purchasing spot and at the same time selling...
-
Draw structures corresponding to the following IUPAC names: (a) 2,3-Dichloro-4-methylhexane (b) 4-Bromo-4-ethyl-2-methylhexane (c) 3-Iodo-2,2,4,4-tetramethylpentane
-
Predict the product(s) of the following transformations: (a) (c) CHOH Periodinane CH3 HC=CHCHCHCOCH 1. LIAIH4 2. HO ? ? (b) (d) CH3 I OCHCHCH3 HC-CHCHCH3 HBr HBr ? ?
-
Niobium forms a substitutional solid solution with vanadium. Compute the weight percent of niobium that must be added to vanadium to yield an alloy that contains 1.55 10 22 Nb atoms per cubic...
-
Goods for which demand is directly (positively) related to income are called ______. a) substitute goods b) complementary goods c) inferior goods d) normal goods
-
Price floors keep prices ___________equilibrium price; price ceilings keep prices ________equilibrium price.
-
If supply is perfectly inelastic, a tax increase is borne _______. a) only by the buyer b) only by the seller c) mostly by the buyer d) mostly by the seller
-
As price rises _____. a) supply rises b) supply falls c) quantity supplied rises d) quantity supplied falls
-
Changes in supply may be caused by changes in _____-. a) the cost of factors of production b) the level of technology c) the number of suppliers d) all of the above e) none of the above
-
Shervonne Thomas is the controller at a large manufacturing company located in Chestereld, Virginia. The company has several divisions that evaluate their performance using a return on investment...
-
Refer to Exercise 8.S.I. Construct a scatterplot of the data. Does the appearance of the scatterplot indicate that the pairing was effective? Explain. Exercise 8.S.I. A volunteer working at an animal...
-
Identify an alkyl halide that could be used to make the following alkene:
-
When menthyl chloride is treated with a strong base, only one elimination product is observed. Yet, when neomenthyl chloride is treated with a strong base, two elimination products are observed. Draw...
-
Predict which of the following two compounds will undergo an E2 reaction more rapidly: CI
-
If you presently have $6,200 invested that will continually earn an annual rate of 4.2%, how long will it take for your investment to triple in value?
-
Formulate a first draft of an elevator pitch for your company. Be sure to address: What exactly does your company do? Why are you and your team qualified to do this? How does your company do what it...
-
Calculate under the assumption that the patients admitted and discharged on the same day and the transfers are NOT newborns. 12:01 a.m. census Adm Trf Total Disch Trf 11:59 p.m. census Serv days Day...

Study smarter with the SolutionInn App


