Three IR spectra are shown, corresponding to three of the following compounds. For each spectrum, determine the
Question:
Three IR spectra are shown, corresponding to three of the following compounds. For each spectrum, determine the structure and explain how the peaks in the spectrum correspond to the structure you have chosen.
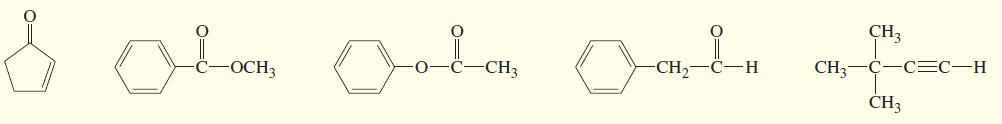
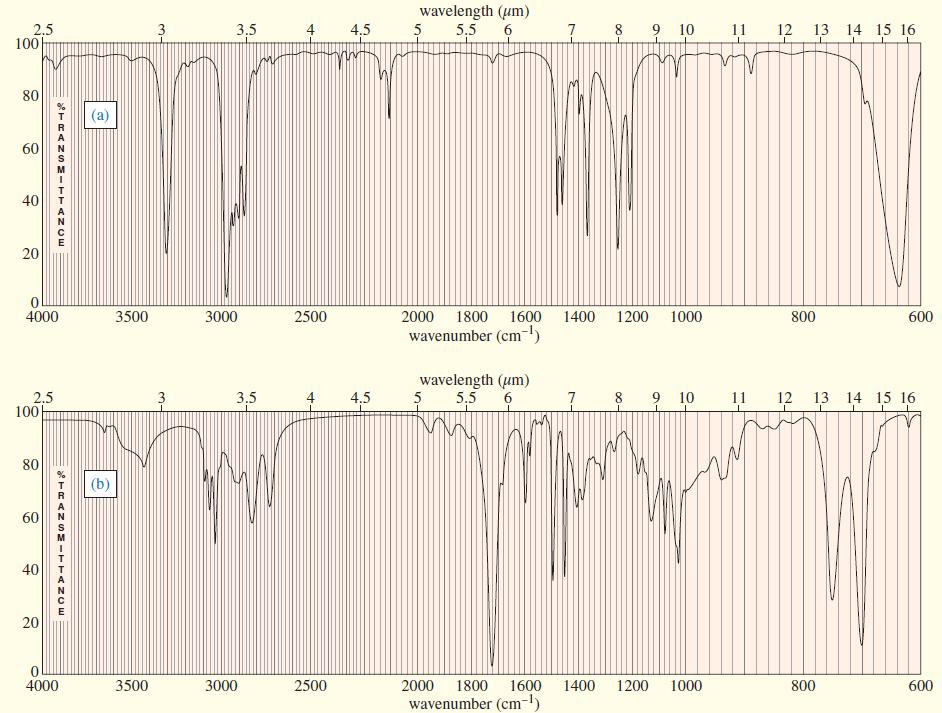
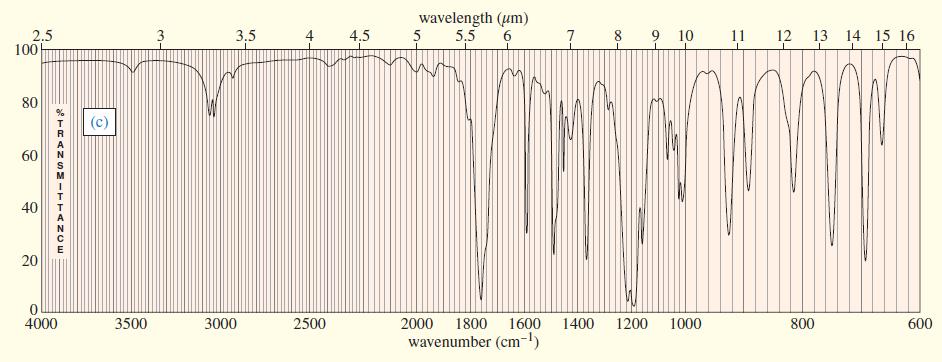
Transcribed Image Text:
CH3 Lon CH CH3 C-C=C-H H. OCH3 CH3 CH3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 70% (10 reviews)

Answered By

Vaibhav Gupta
I am studying in the number 1 university of India i. e Delhi university as a physicist.
And I am also my school and university topper and I am tutoring around 3 years to many students and they are always satisfied from my tutoring.
I will do my best to provide best explanation with answers.
Thankyou
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Four infrared spectra are shown, corresponding to four of the following compounds. For each spectrum, determine the structure and explain how the peaks in the spectrum correspond to the structure you...
-
The 1H NMR spectrum shown in Figure 14.8 corresponds to one of the following compounds. Which compound is responsible for this spectrum? C CH CH3 CH3 CICH2 CH2C Br2CH CHBr2 6 5 3 2 0 8 (ppm)...
-
The IR spectrum shown in Figure 13.43 is the spectrum of one of the following compounds. Identify the compound. CH2OH COOH CH2NH2 3 2.6 27 28 20 3 12 13 14 5 6 4003360 3400 3200 3000 2800 2800 2400...
-
Compute the given derivatives with the help of formulas (1)(4). (a) (b) d dx (1) - x=e
-
A 0.0450 M solution of HA is 0.60% dissociated. Calculate pKa for this acid.
-
Which of the following would you expect to be strong electrolytes when placed in water? NH 4 Cl MgBr 2 H 2 O HCl Ca 3 (PO 4 ) 2 CH 3 OH Which of the statements below support the answers you chose...
-
You borrow \($495\) from your brother-in-law to buy an iPad and agree to pay it back when you get your tax refund (6 months away). Because you are family, but not trusted, you are being charged...
-
A 2 mol% NH3-in-air mixture at 68F and 1 atm is to be scrubbed with water in a tower packed with 1.5-in, ceramic Berl saddles. The inlet water mass velocity will be 2400 lbk-ft2, and the inlet gas...
-
Given the following probability distribution for Foster Trucking Company Possible outcome for damages Probability Calculate the expected value. $0 0.4 $400 0.3 $1,000 0.2 $5,000 0.1
-
Snyder Computer Chips, Inc., is experiencing a period of rapid growth. Earnings and dividends are expected to grow at a rate of 15 percent during the next two years, at 13 percent in the third year,...
-
Chapter 9 covered a synthesis of alkynes by a double dehydrohalogenation of dihalides. A student tried to convert trans-2,5-dimethylhex-3-ene to 2,5-dimethylhex-3-yne by adding bromine across the...
-
A laboratory student added 1-bromobutane to a flask containing dry ether and magnesium turnings. An exothermic reaction resulted, and the ether boiled vigorously for several minutes. Then she added...
-
What difficulties arise in the polymerase chain reaction if there is contamination of the DNA that is to be copied?
-
Should you use the direct or indirect approach for most routine messages? Why?
-
You work for an importexport business that has, until recently, only offered services to other businesses. Recently, you noticed the opportunity to make full use of shipping containers by offering...
-
Imagine that you are the president of the campus business debating and presentation group. You routinely book business professionals who have something interesting or thought-provoking to say. The...
-
Between this chapter and your own experience as a user of social media, you know enough about social media to offer some insights to other business communicators. Your task: Write a script for a two-...
-
Produce a tabulated list of advantages and disadvantages of using the following list of secondary research sources to produce a valid and reliable report: a. The annual report of a large company b. A...
-
Maple Leaf Foods Inc. reported the following selected information (in thousands): _________________________________20112010 Total assets...............................$2,940,459...........$2,834,910...
-
A line l passes through the points with coordinates (0, 5) and (6, 7). a. Find the gradient of the line. b. Find an equation of the line in the form ax + by + c = 0.
-
How would the proton NMR spectrum of the com-pound in part (a) change following a D2O shake?
-
Imagine taking the NMR spectrum of a sample of "naked" protons-that is, H+ in the gas phase not chemically bound to anything. In which of the following ranges of chemical shifts would you expect to...
-
The gyromagnetic ratio of the electron is 17.60 106 rad gauss-1 s-1, 658 times greater than that of the proton. What operating frequency would be required to detect the magnetic resonance of an...
-
Of 15,000 individuals aged 18 years living in Ontario, 5,000 visited their family doctor in the past year and of these individuals 1,875 were diagnosed with lifelong depression. Assuming everyone was...
-
If A = 9 3 -5 -8 -7 01-87 2 00-765 000-4 3 0 0 0 0 -9 then det (A) =
-
To improve the effectiveness of its teaching staff, the administration of a high school offered the opportunity for all teachers to participate in a workshop. They were not required to attend;...

Study smarter with the SolutionInn App


