Where would you expect to find the 1 H NMR signal of (CH 3 ) 2 Mg
Question:
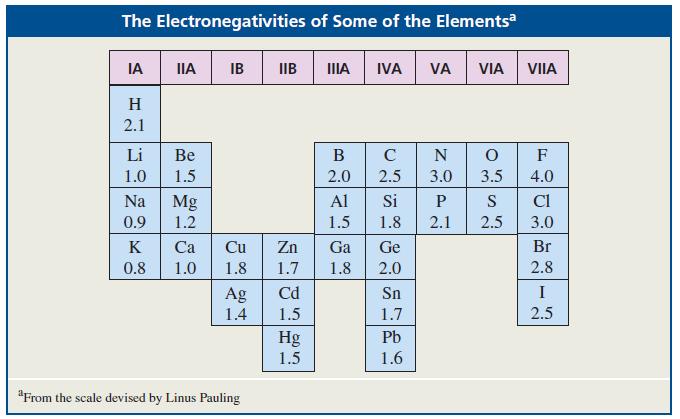
Where would you expect to find the 1H NMR signal of (CH3)2Mg relative to the TMS signal? (See Table 12.3.)
Table 12.3
Transcribed Image Text:
The Electronegativities of Some of the Elementsa IA IIA IB IIB А IVA VA VIA VIIA H 2.1 Li Ве В C N F 1.0 1.5 2.0 2.5 3.0 3.5 4.0 Na Mg Al Si P S CI 0.9 1.2 1.5 1.8 2.1 2.5 3.0 K Са Cu Zn Ga Ge Br 0.8 1.0 1.8 1.7 1.8 2.0 2.8 Cd I Ag 1.4 Sn 1.5 1.7 2.5 Pb Hg 1.5 1.6 "From the scale devised by Linus Pauling
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 57% (14 reviews)
Because Mg is less electronegative than Si the methy...View the full answer

Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
In what chemical shift ranges would you expect to find the proton NMR signals of ethyl acetate (CH3CO2CH2CH3)?
-
How many signals would you expect to find in the 1H NMR spectrum of each of the following compounds? (a) 1-Butanol (b) Butane (c) 1,4-Dibromobutane
-
(a) How many signals would you expect to find in the 1H NMR spectrum of caffeine? (b) What characteristic peaks would you expect to find in the IR spectrum of caffeine? CH3 CH3 Caffeine
-
Refer to the RMO CSMS Order Fulfillment subsystem shown in Figure. Draw a use case diagram that shows all actors and all use cases. Use a drawing tool such as Microsoft Visio if it is available.
-
The exitance (power per unit area per unit wavelength) from a blackbody (Box 19-1) is given by the Planck distribution: where _ is wavelength, T is temperature (K), h is Planck's constant, c is the...
-
You are attending a student engineering organization regional conference along with five other students from your institution. The night before the group is scheduled to return to campus, one of the...
-
The following information was reported in the December 31, 2007, financial statements of Southwest Airlines, Inc. (listed alphabetically, amounts in millions). Accounts Payable $1,731 Accounts...
-
Refer to Case 2-1 above. Mr. Pitt has relented and agrees to provide you with the information necessary to convert his cash basis financial statements to accrual basis statements. He provides you...
-
Prepare journal entries for the following transaction using the accrual basis of accounting. Received $2,000 from a charge client for services performed last month. For the toolbar, press ALT+F10...
-
1. Jim Adams posts the column totals and individual transactions in "Other" columns to the appropriate general ledger account and updates each general ledger account balance. He places a posting...
-
How could you distinguish the 1 H NMR spectra of the following compounds? a. CH 3 OCH 2 OCH 3 b. CH 3 OCH 3 c. CH3 CH3OCH,CCH2OCH3 H3
-
a. Calculate the ratios of the different kinds of protons in a compound with an integral ratio of 6 : 4 : 18.4 (going from left to right across the spectrum). b. Determine the structure of a compound...
-
Compare the retail sale of 10,000 feet of irrigation pipe from a Farmers Supply to a farmer with the commercial sale of that same amount of pipe from a wholesaler to the Farmers Supply. Label the...
-
Discuss the relevance of social media marketing to B2B organisations.
-
Explain the different types of payment model for banner advertising.
-
Describe three alternative locations for transactions for a B2B company on the Internet.
-
Discuss the relative effectiveness of the different methods of assessing the customers needs from a website.
-
Discuss why a business operating in an industrial market might be cautious about putting new product specifications on the company website.
-
What types of cash flows does a bond buyer receive?
-
For the vector whose polar components are (Vr = 1, Vθ = 0), compute in polars all components of the second covariant derivative Vα;μ;ν. To find...
-
Give Lewis structures corresponding to the following line-angle structures. Give the molecular formula for each structure. (a) (b) (c) (d) (e) (f) (g) (h) OH CHO
-
Repeat Problem 1-9, this time drawing line-angle structures for compounds (a) through (h). In problem 1-9 Draw complete Lewis structures for the following condensed structural formulas. (a)...
-
Compute the empirical and molecular formulas for each of the following elemental analyses. In each case, propose at least one structure that fits the molecular formula. C1 (a) (b) (c) (d) 40.0% 32.0%...
-
There are 42 locations throughout the northeast region with deposits totaling $3.2bil. If you were Kiamarie's vice president, how would you expect her to choose between Gagne's Nine Events of...
-
Why is succession planning essential in an organization? 2. What is the need for succession planning? 3. What do you think are the challenges to succession planning? 4. How can employees contribute...
-
Two activities/obligations/responsibilities that might be added to an updated version of the job description for an HR professional: How would you go about monitoring changes in laws and regulatory...

Study smarter with the SolutionInn App


