Which of the following are likely to act as Lewis acids and which as Lewis bases? Which
Question:
Which of the following are likely to act as Lewis acids and which as Lewis bases? Which might act both ways?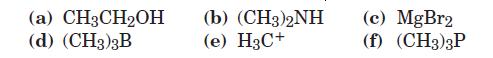
Transcribed Image Text:
(a) CH3CH2OH (d) (CH3)3B (b) (CH3)2NH (e) H3C+ (c) MgBr2 (f) (CH3)3P
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 16% (6 reviews)
In the context of Lewis acids and bases Lewis acids are substances that can accept a pair of electrons to form a new covalent bond Lewis bases are sub...View the full answer

Answered By

Saleem Abbas
Have worked in academic writing for an a years as my part-time job.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Which of the following are Lewis acids and which are Lewis bases? a. Mg2+ b. (CH3)3C:- c. CH3NH2 d. Zn2+ e. CH3CH2OCH2CH3 f. (CH3)3C+ g. (CH3)3B h. (CH3)3N i. H:-
-
Proteins are synthesized with a particular amino acid sequence through the translation of information encoded in messenger RNA by an RNAprotein complex called a ribosome. Amino acids are specified by...
-
Show how the species in part (a) can act as Lewis bases in their reactions with HCl, and show how the species in part (b) can act as Lewis acids in their reaction with OH - . (a) CH3CHOH, (CH3)2NH,...
-
Calculate the labour turnover rate according to replacement method from the following: No. of workers on the payroll: - At the beginning of the month: 500 - At the end of the month: 600 During the...
-
Following are the adjusted current funds trial balances of Community Association for Children With Disabilities, a voluntary health and welfare organization, on June 30, 20X4: Required a. Prepare a...
-
What is white space and why is it important for readability?
-
Amanda Forbes was hired as a nail technician by Showmann, Inc., in 2011. In 2017, Forbes attended a work-related holiday party where Showmann distributed raffle tickets to employees. One of the...
-
1. Suppose you belong to a tennis club that has a monthly fee of $100 and a charge of $5 per hour to play tennis. a. Using Figure as a model, prepare a table and draw a curve to show the relationship...
-
1/ You have two different assets (investments). Asset A (perpetuity) will pay you $1,000 in one year, $1,000 in two years, $1,000 in three years, and so on every year forever. Asset B will pay you...
-
Convert the following line-bond structures into molecular formulas: (a) (b) (c) . CHOH . C C C=C OH Vitamin C (ascorbic acid)
-
Electrostatic potential maps of (a) acetamide and (b) methylamine are shown. Which of the two has the more basic nitrogen atom? Which of the two has the more acidic hydrogen atoms? (a) (b) Acetamide...
-
Japan has typically had lower inflation than the United States. How would one expect this to affect the Japanese yens value? Why does this expected relationship not always occur?
-
A charge (q=-2 C) is next to (but not inside) an enclosed surface in the shape of a cube. What is the electric flux through the surface?
-
Design and develop Card, Deck, and Tableau classes The Card class (20 marks) represents a playing card, where each has a rank (2, 3, , 9, T, J, Q, K, and A) and a suit (C, D, H, and S) field. When...
-
A baseball player wants to hit a home run over the wall of a stadium. The player swings the baseball bat so that it hits the ball when it is at a height of 1.19 m above the ground. The ball flies off...
-
An agency increased its monthly expenditures by 25% from January 2009 to January 2010. During January 2010, the agency's monthly expenditures totaled $2,050. During February 2010, monthly...
-
If a 2 A current flows in a resistor and this resistor produces a voltage drop of 10 V. What is the power dissipated by the resistor?
-
As an investment advisor, you tell a client that an investment in a mutual fund has (over the next year) a higher expected return than an investment in the money market. The client then asks the...
-
Time Travel Publishing was recently organized. The company issued common stock to an attorney who provided legal services worth $25,000 to help organize the corporation. Time Travel also issued...
-
What two monosaccharides can be degraded to a. D-arabinose? b. D-glyceraldehyde? c. L-ribose?
-
Aldohexoses A and B form the same osazone. A is oxidized by nitric acid to an optically active aldaric acid, and B is oxidized to an optically inactive aldaric acid. Ruff degradation of either A or B...
-
What kind of aldohexose would form L-glyceraldehyde when its acetal is oxidized with periodic acid?
-
In (Figure 1), C = C5 = 8.6 F and C = C3 = C = 4.8 F. The applied potential is Vab = 210 V. What is the equivalent capacitance of the network between points a and b? Express your answer in farads....
-
Two charges +Q and -9Q are placed on the x axis with x coordinates=2m and 8m respectively. Find the coordinate of a point on the x axis between the two charges at which the potential is zero....
-
Katz establishes that the Fourth Amendment regulates some surveillance beyond physical intrusion. But surveillance is covered? Is the key question whether the surveillance invades shared senses of...

Study smarter with the SolutionInn App


