(a) Aliphatic allylic vinylic ethers undergo the Claisen rearrangement. Complete the following reaction: (b) What starting material...
Question:
(a) Aliphatic allylic vinylic ethers undergo the Claisen rearrangement. Complete the following reaction:
![]()
(b) What starting material would give the following compound in an aliphatic Claisen rearrangement?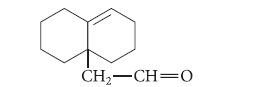
Transcribed Image Text:
(CH;),C=CH–CH, O–CH=CH, heat
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 40% (5 reviews)
a Deduce the product by using the curvedarrow ...View the full answer

Answered By

Gaurav Soni
Myself a student of Physics currently pursuing my PG. I helped many students due to my understanding specifically among students and also in my subject i. e. Physics. Helping them in various boards and in various entrance examination gives me a satisfaction of gaining knowledge. I have taught physics to students for many engineering and medical entrance examination and also for various boards in india. Till now have a great results from my students and that's why having a good impact on them. I have a good knowledge in conceptual and theoretical physics. Essential University Physics is one of my favorite field of teaching. I have a experience of almost 4 years in teaching.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
What starting material would give the following compound in an aliphatic Claisen rearrangement? CH,-CH-C
-
The mechanism of the Claisen rearrangement of other allylic ethers of phenol is analogous to that of allyl phenyl ether. What is the product of the Claisen rearrangement of C6H5OCH2CH==CHCH3?
-
The mechanism of the Claisen rearrangement of other allylic ethers of phenol is analogous to that of allyl phenyl ether. What is the product of the Claisen rearrangement of C6H5OCH2CH=CHCH3?
-
You have observed the following returns over time: Assume that the risk-free rate is 6% and the market risk premium is 5%. a. What are the betas of Stocks X and Y? b. What are the required rates of...
-
A recent annual report for FedEx includes the following information: For financial reporting purposes, we record depreciation and amortization of property and equipment on a straight-line basis over...
-
What is treasury stock and why is it acquired?
-
The John Gore Organization owns and operates the Charles Playhouse, a theater in Boston, Massachusetts. Evelyn Castillo has diabetes, a disability under the Americans with Disabilities Act (ADA)....
-
Bill Fennema, president of Fennema Hospitality, has developed the tasks, durations, and predecessor relationships in the following table for building new motels. Draw the AON net-work and answer the...
-
A child bounces a 48 g superball on the side- walk. The velocity change of the superball is from 23 m/s downward to 13 m/s upward. If the contact time with the sidewalk is 1 800 s, what is the...
-
Show how the transition state for a [3,3] sigmatropic reaction can be analyzed as the interaction of two allylic radicals, and that the same stereochemical outcome is predicted.
-
(a) What allowed and reasonable sigmatropic reaction(s) can account for the following transformation? (b) What product(s) are expected from a similar reaction of 2,3-dimethyl-1,3- cyclopentadiene?...
-
What effect would each of the following, taken alone, have on a federal budget that is currently balanced? How would the budgetary changes, where they occur, affect the national debt? a. An increase...
-
The goal is to make a digital representation of a car that can later be physically constructed. The primary requirement is for the car to move forward, without the need for any additional...
-
A box of mass m=25 kg is pushed along a rough surface by a force applied of magnitude 40 N directed at 0=45 degrees below horizontal. The coefficient of kinetic friction between the box and the...
-
Consider a 1-year zero-coupon bond with $1,000 face value. Assume today's 6-monthforward rate is 6% and in six months the new 6-month forward rate can be either 5% or 7%. Assume also that the current...
-
Calculate the total heat gains of the window below Dimension: 9' by 13' Materials: 1" insulating glass, 1/4" lites, clear (see Table 3.5) Note: SC = 0.23 due to blinds Orientation: South Time: When...
-
An extraction column is used to remove an impurity P from toluene with water as the solvent. The toluene and water may be considered to be completely immiscible. The toluene enters the column with 20...
-
What is the inventory equation?
-
What is the back work ratio? What are typical back work ratio values for gas-turbine engines?
-
For which of the following ions does the formal charge give a fairly accurate picture of where the charge really is? Explain in each case. (a) NH4 (b) H3O: (C) NH2 (d) CH3
-
Is there an unbranched alkane containing 23 hydrogen atoms? If so, give its structural formula; if not, explain why not.
-
In the structure of 4- isopropy 1-2,4,5-trimethylheptane (Problem 2.9) (a) Identify the primary, secondary, tertiary, and quaternary carbons. (b) Identify the primary, secondary, and tertiary...
-
"It's "Virtually" the Same Game?! The business of sports is growing tremendously and it is fueled in part by athletes that take on celebrity status. None typifies this better than Tiger Woods. Tiger...
-
Why should Maersk's human resource management align with the organizations strategic plan
-
Introduce the topic of algorithms in the selection process. How might the recommendations an algorithm makes differ from those of a hiring manager who is not using data analytics?

Study smarter with the SolutionInn App


