A box labeled C 6 H 10 isomers contains samples of three compounds: A, B, and C.
Question:
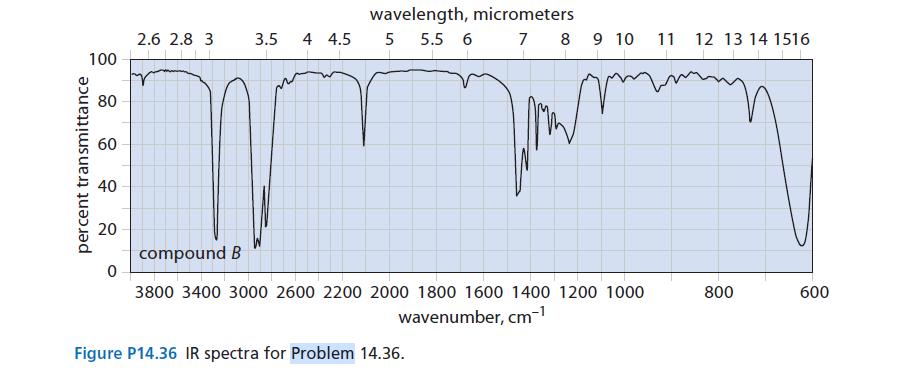
A box labeled “C6H10 isomers” contains samples of three compounds: A, B, and C. Along with the compounds are the IR spectra of A and B, shown in Fig. P14.36. Fragmentary data in a laboratory notebook suggest that the compounds are 1-hexyne, 2-hexyne, and 3-methyl-1,4-pentadiene. Identify the three compounds.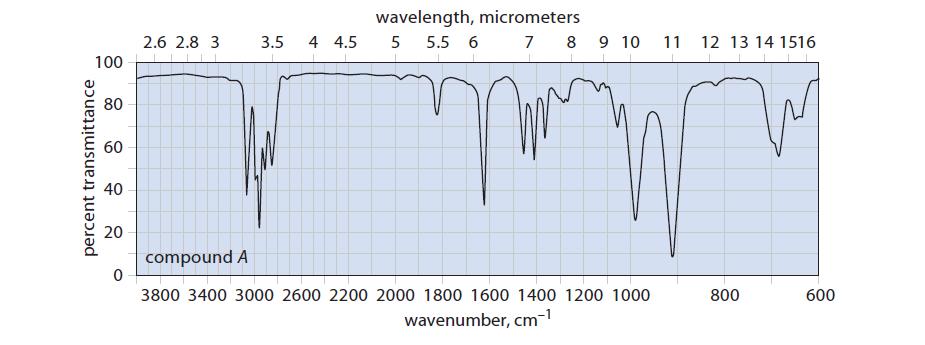
Transcribed Image Text:
percent transmittance 100 80 60 40 20 0 2.6 2.8 3 3.5 4 4.5 wavelength, micrometers 5 5.5 6 7 8 9 10 11 12 13 14 1516 m compound A 3800 3400 3000 2600 2200 2000 1800 1600 1400 1200 1000 wavenumber, cm-¹ 800 600
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
First look for the telltale CH and CC stretching absorpt...View the full answer

Answered By

Aqib Parvej
I am teaching since my graduation time so I have teaching experience of about 5 years and in these years I learn to teach in the best and interesting way .
4.80+
20+ Reviews
41+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-5. Ivan's grandfather died and left a portfolio of municipal bonds. In 2012, they pay Ivan...
-
The IR and mass spectra for three different compounds are shown in Figures 13.45-13.47. Identify each compound. a. b. c. 25 26 2.7 28 19 2000 100 43 58 100 71 85 0 20 40 60 80 100 120 m/z 13 14 15 6...
-
The following additional information is available for the Dr. Ivan and Irene Incisor family. The Incisors own a rental beach house in Hawaii. The beach house was rented for the full year during 2012...
-
There are several problems that are unique to family businesses or ventures. Which of the following is not one of those problems? Issues of succession Problems of control, fairness, and equity Issues...
-
Carters Produce has applied for a loan and has agreed to use its inventory to collateralize the loan. The company currently has an inventory balance of $289,000. The cost of goods sold for the past...
-
Milano Pizza Club owns three identical restaurants popular for their specialty pizzas. Each restaurant has a debtequity ratio of 40 percent and makes interest payments of $41,000 at the end of each...
-
On April 4, 2014, Athanasios Valsamis lost his appeal to get his money back from a friend to whom he had loaned \($700,000.\) As you will read, this case underscores the consequence of failing to...
-
Cost Structure to erratic sales of its sole producta high-capacity battery for laptop computersPEM, Inc., has been experiencing difficulty for some time. The companys contribution format income...
-
explain diagram of ER model and relational model and attach their diagrams too Each cinema is identified by its name and has its residency at an address which consists of a street and city only....
-
(a) Draw the structures of all enols that would spontaneously form the following ketone, including stereoisomers. (b) Would alkyne hydration be a good preparative method for this compound? If so,...
-
Give the principal product(s) expected when 1-hexyne or the other compounds indicated are treated with each of the following reagents: (a) HBr (b) H, Pd/C (c) H, Pd/C, Lindlar catalyst (d) product of...
-
An article in the New York Times has the headline "U.S. Trade Deficit Widened Over Summer." The first sentence in the article is, "The current-account trade deficit widened slightly in the...
-
Suppose the exchange rate between the U.S. dollar and the Swedish krona was 8.8 krona = $1, and the exchange rate between the dollar and the British pound was 1 = $1.3. What would be the exchange...
-
Early in June 1983, it took 245 Japanese yen to equal $1. In August 2020, that exchange rate had fallen to 106 yen to $1. Assume that the price of a Japanese-manufactured automobile was $9,000 in...
-
You are the vice president of Worldwide InfoXchange, headquartered in Minneapolis, Minnesota. All shareholders of the firm live in the United States. Earlier this month, you obtained a loan of 10...
-
Describe the way post-merger cash flows are estimated in a DCF analysis. What is the basis for the discount rate in a DCF analysis? Describe how this rate might be estimated.
-
Give an example of an if-then statement. Why might using causal research enhance the accuracy of your findings?
-
Fatima inherits a rental property with a fair market value of $90,000 from her aunt on April 30. On May 15, the executor of the estate sends her a check for $7,000. A letter accompanying the check...
-
"Standard-cost procedures are particularly applicable to process-costing situations." Do you agree? Why?
-
Would you expect a conjugated diyne such as 1, 3-butadlyne to undergo DielsAlder reaction with a dienophile explain.
-
Reaction of isoprene (2-methyl-1, 3-hutadiene) with ethyl propenoate gives a mixture of two Diels?Alder adducts. Show the structure of each, and explain why a mixture is formed. CO2CH2CH3
-
Rank the following dienophiles in order of their expected reactivity in the Diels?Alder reaction. CH CH NC NC CN c=C CH c=c c=C c=C NC CN
-
The English alphabet has 26 letters, of which 5 are vowels and 21 are consonants. We will write all our words using upper case (capital) letters. Repetition of letters in words is allowed. Find the...
-
General Products writes checks that average $28,000 daily. These checks take an average of 6 days to clear. It receives payments that average $30,000 daily. It takes 3 days before these checks are...
-
In Drosophila, white eyes are an X-linked character. The mutant allele for white eyes (w) is recessive to the wild-type allele for brick red eye color (w). A white-eyed female is crossed with a...

Study smarter with the SolutionInn App


