(a) Draw the structures of all enols that would spontaneously form the following ketone, including stereoisomers. (b)...
Question:
(a) Draw the structures of all enols that would spontaneously form the following ketone, including stereoisomers.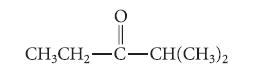
(b) Would alkyne hydration be a good preparative method for this compound? If so, give the reaction. If not, explain why.
Transcribed Image Text:
CH3CH₂-C-CH(CH3)2
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
a b Two constitutionally isomeric enols are possible one of which ...View the full answer

Answered By

Asim farooq
I have done MS finance and expertise in the field of Accounting, finance, cost accounting, security analysis and portfolio management and management, MS office is at my fingertips, I want my client to take advantage of my practical knowledge. I have been mentoring my client on a freelancer website from last two years, Currently I am working in Telecom company as a financial analyst and before that working as an accountant with Pepsi for one year. I also join a nonprofit organization as a finance assistant to my job duties are making payment to client after tax calculation, I have started my professional career from teaching I was teaching to a master's level student for two years in the evening.
My Expert Service
Financial accounting, Financial management, Cost accounting, Human resource management, Business communication and report writing. Financial accounting : • Journal entries • Financial statements including balance sheet, Profit & Loss account, Cash flow statement • Adjustment entries • Ratio analysis • Accounting concepts • Single entry accounting • Double entry accounting • Bills of exchange • Bank reconciliation statements Cost accounting : • Budgeting • Job order costing • Process costing • Cost of goods sold Financial management : • Capital budgeting • Net Present Value (NPV) • Internal Rate of Return (IRR) • Payback period • Discounted cash flows • Financial analysis • Capital assets pricing model • Simple interest, Compound interest & annuities
4.40+
65+ Reviews
86+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The following compound was isolated from nerve cells. (a) Describe how this compound differs in structure from fats and oils. (b) Three products are obtained when this compound is hydrolyzed with...
-
Draw the structures of all compounds with the formula C6H12C12 that can exist as meso compounds. Indicate how many meso compounds are possible for each structure.
-
Give the structures of all epoxides that could in principle be formed when each of the following alkenes reacts with meta-chloroperoxybenzorc acid (mCPBA). Which epoxide should predominate in each...
-
Question 3 A bullish signal is most likely sent by B a low debt/equity ratio. A a high P/E ratio. B a high good beta and a low bad beta. Question 4 a bullish candlestick with no upper shadow. How to...
-
The following accounting information pertains to Clemens Corp. and Twain Inc. at the end of 2009. The only difference between the two companies is that Clemens uses FIFO while Twain uses LIFO....
-
Gemini, Inc., an all-equity firm, is considering a $1.7 million investment that will be depreciated according to the straight-line method over its four-year life. The project is expected to generate...
-
T. Christian Cooper was a partner to Sanders and Richard Campbell d/b/a The Mullen Company. In 2001, Cooper helped bring about a management agreement between The Mullen Co. and Newnan Crossing...
-
Select the graph that matches the numbered manufacturing cost data. Indicate by letter which graph best fits the situation or item described. The vertical axes of the graphs represent total cost and...
-
To determine the kinetics (rates) of ozone depletion reactions, chemists perform controlled laboratory studies. In this simulated lab, we will interpret data obtained from such laboratory experiments...
-
In each case, draw a structure containing only carbon and hydrogen that satisfies the indicated criterion. (a) A stable alkyne of five carbons containing a ring (b) A chiral alkyne of six carbon...
-
A box labeled C 6 H 10 isomers contains samples of three compounds: A, B, and C. Along with the compounds are the IR spectra of A and B, shown in Fig. P14.36. Fragmentary data in a laboratory...
-
Apple Inc. is an international corporation that designs, manufactures, and markets a range of mobile communication and media devices, personal computing products, and portable digital music players,...
-
Why are bond ratings important to firms and investors?
-
Does the average investors willingness to take on risk vary over time? Explain.
-
Should companies completely avoid high-risk projects? Explain.
-
Have there been any studies that question the validity of the CAPM? Explain.
-
What do you think the average investors risk perception is now? In what types of investments do you think the average investor is investing currently?
-
Allison dies during the current year. She is covered by a $1,000,000 life insurance policy payable to her husband, Bob. Bob elects to receive the policy proceeds in 10 annual installments of...
-
The Zwatch Company manufactures trendy, high-quality moderately priced watches. As Zwatch's senior financial analyst, you are asked to recommend a method of inventory costing. The CFO will use your...
-
Predict the products of the following Diels?Alder reactions; (b) (a)
-
Show the structure, including stereochemistry, of the product from the following Diels?Alder reaction: COCH3 C
-
How can you account for the fact that cis-1, 3-pentadicne is much less reactive than trans-1, 3-pentadiene in the DielsAlder reaction?
-
The project will teach you how to perform a financial analysis on a set of financials. A financial analysis report is constructed by a person who is researching a company, usually with the intent of...
-
Orchard B&B is a bed and breakfast on the coast of Florida. As customers make reservations they are required to pay cash in advance equal to one-half of the rate for their stay. How should Orchard...
-
Following are three economic states, their likelihoods, and the potential returns. Determine the standard deviation of the expected return. (Do not round intermediate calculations and round your...

Study smarter with the SolutionInn App


