(a) Draw the structure of a phosphotyrosine residue. (b) Would the equilibrium constant for formation of a...
Question:
(a) Draw the structure of a phosphotyrosine residue.
(b) Would the equilibrium constant for formation of a phosphotyrosine residue from ATP (by a reaction analogous to the one shown in Eq. 27.43) be greater than, less than, or about the same as Keq for the phosphorylation of a serine residue? Explain.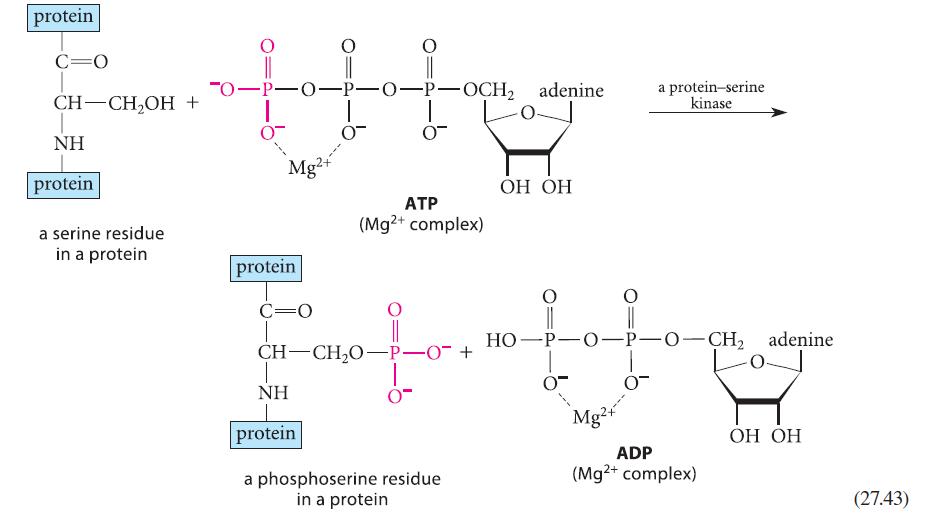
Transcribed Image Text:
protein C=O CH-CH₂OH NH protein a serine residue in a protein + -O-P-O-P-O-P-OCH₂ adenine 0 Mg2+ protein C=O T NH protein 0 ATP (Mg2+ complex) CH-CH₂O-P-0 + 0- a phosphoserine residue in a protein OH OH HO-P-O-P-O-CH₂ adenine Mg a protein-serine kinase 0 ADP (Mg²+ complex) OH OH (27.43)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
a Fot CHCH 01P10 a phosphotyrosine residue in a protein NH b T...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Draw the structure of each of the following. (Some parts may have more than one correct answer.) (a) A nine-carbon ether that can be prepared by the Williamson synthesis. (b) An ether that would...
-
A graduate student was following a procedure to make 3-propylcyclohexa-1,4-diene. During the workup procedure, his research adviser called him into her office. By the time the student returned to his...
-
Draw the structure of the product from the following reaction (formed during a synthesis of one of the endiandric acids by K. C. Nicolaou): MeO2C osi(t-Bu)Phe toluene, 110C
-
1. Insurance Act, RSBC 1996 c226 Read Parts 1 and 2 of this statute and describe any changes to the standard commonlaw rules for contracts that you notice. 2. KP Pacific Holdings Ltd. and Churchland...
-
In this chapter, we discussed the importance of analyzing financial results based on an understanding of the companys business strategy. Using the ROE model, we illustrated how different strategies...
-
What are American Depositary Receipts (ADRs), and why have they proven so popular with U.S. investors?
-
A project has been selected for implementation. The net cash flow (NCF) profile associated with the project is shown below. MARR is 10 percent/year. a. What is the internal rate of return of this...
-
(One Temporary Difference, Tracked for 4 Years, One Permanent Difference, Change in Rate) The pretax financial income of Truttman Company differs from its taxable income throughout each of 4 years as...
-
Q3. If W and W are two independent Brownian motions and p is a constant between -1 and 1, then the process Xt = pWt + 1 pWt is continuous and has - marginal distributions N(0, t). Is this X a...
-
(a) Draw a resonance structure for the carbocation intermediate in Eq. 27.48. (b) All of the enzyme-catalyzed glycosylations require Mn 2+ , a divalent cation. Suggest a role for the metal ion in the...
-
Some peptides found in nature have an amino-terminal acetyl group (red): (a) Can these peptides undergo the Edman degradation? Explain. (b) Does N-acylation have any adverse effect on sequencing by...
-
What does product life cycle mean?
-
Assumptions : Sales will increase by 15% COGS will increase by 150 Basis Points General and Admin Expenses will Increase by 50 basis points R & D, Current Assets and Current Liabilities will remain...
-
1. Why do you think the certificate option is best for me compared to the Google PM Certificate. 2. Develop a timeline with the main milestones to get this certificate (If you need to gain experience...
-
Identify specific accounts you would specifically want to spend your audit time on due to higher Risk of Material Misstatement (RMM). LIABILITIES AND SHAREHOLDERS EQUITY: Current liabilities:...
-
1. What are post-project reviews? Why are they necessary? 2. What are the eight major problems that act as barriers to learning from post-project reviews? 4. What are the benefits of audits in...
-
From the Notes to the Financials: NIKE lululemon (For the most current year reported) Allowance for uncollectible/doubtful accounts receivable Increase or decrease over prior year end? Inventory...
-
What are direct and indirect costs?
-
Assume Eq. 6-14 gives the drag force on a pilot plus ejection seat just after they are ejected from a plane traveling horizontally at 1300 km/h. Assume also that the mass of the seat is equal to the...
-
Indicate whether each of the following statements is true or false. If false, explain why. (a) In every case, pair of enantiomers have a mirror- image relationship. (b) If a compound has an...
-
Draw the structures of all compounds with the formula C6H12C12 that can exist as meso compounds. Indicate how many meso compounds are possible for each structure.
-
Identify the asymmetric carbon(s) (if any) in each of the following molecules. ,
-
Find the missing length of the triangle 40 75 Feet a) 62.9 feet b) 48.2 feet c) 57.5 feet d) 72.2 feet
-
Suppose 1 an converges. Prove that lim r-1, r <1 n=1 ran = An. n=1
-
Problem Solving An MP3 player Cost R2699. How much will R796 MP3 player cost? A charity organisation Collects 628 boxes of gifts. The content of each box is worth R5250. What is the Value of all the...

Study smarter with the SolutionInn App


